We get a fair number of questions here in the forum about whether you should use generic versions of Rapamycin (the brand drug is Pfizer Rapamune), as well as questions about quality, and what has been approved in different countries, etc, and transitionining to different versions, etc.
So when this recent paper came out, I reviewed it and found information in it that people may find interesting and answer some of their questions related to the aforementioned issues.
The paper is from a congenial group of researchers in Poland who were nice enough to send me a copy of their paper. While this is obviously targeted more at transplant patients, lots of information is relevant to anyone interested in rapamycin use.
Following are some sections of the paper I found interesting and perhaps relevant to people here.
Generic Medicinal Products in Immunosuppressive Therapy—Should It be a Challenge for Therapeutic Drug Monitoring?*
It is worth noting that considering the current guidelines, only AUC and Cmax are decisive parameters for proving bioequivalence. It might seem that because the AUC parameter reflects the amount of drug absorbed, it is the most important actor in bioequivalence, but it is not entirely true. Of interest, Cmax had a much greater influence on the success or failure of bioequivalence. It has been proven that Cmax generally shows higher (even several times) interindividual variability than the AUC parameter: bioequivalence is not achieved, and it is often because of Cmax.5,10 During standard crossover bioequivalence studies, 2 pharmacokinetic profiles were obtained for each participant: one after administration of the comparator (original drug) and the other after administration of the potential generic. Sometimes, when the study concerned substances classified as so-called highly variable medicinal products (HVDP) presenting great (.30% CV) intraindividual variability in pharmacokinetic parameters, the study periods were doubled, and 4 pharmacokinetic profiles for one study subject were collected. Such an operation isacceptable and allows us to opt out of a large group of participating volunteers for statistical reasons. In addition, itis worth noting to the fact that the bioequivalence investigation between original and generic formulation is conducted only in the group of healthy volunteers. Some transplant communities have been appealed that bioequivalence investigation of generic NTIDs needs alternative criteria of selecting participants for trials,namely, including transplant recipients in that testing. A special attention should be paid for differences in ethnic, age, sex, and comorbidities of recipients.
…
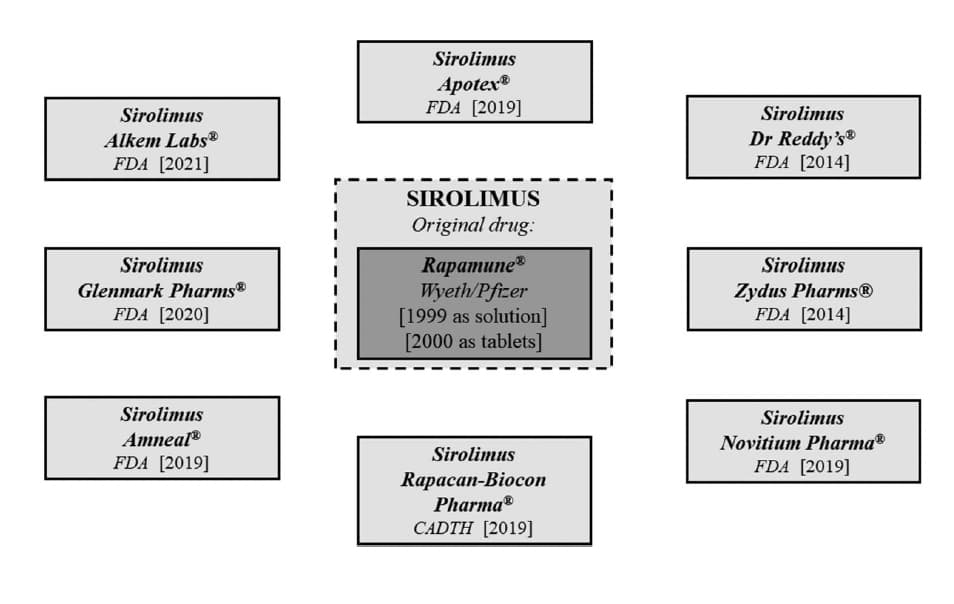
SIR (Sirolimus, Rapamycin)
Sirolimus was introduced to therapy under Rapamune name first in 1999 as an oral emulsion, and 1 year later as tablets by Wyeth (now part of the Pfizer company). Because original formulations containing SIR have been introduced for treatment relatively recently, there are few generics available in the pharmaceutical market. Currently, only the FDA and CADTH agencies have introduced a few SIR generic products. In the United States, 2 of these were introduced by Dr Reddy’s Inc and Zydus Pharma Inc in 2014. The situation is different in Canada, where only one generic SIR was introduced by Rapacan-Biocon Pharmaceuticals Inc in 2019. Recently, 5 new generics were approved by the FDA, and it seems that the number of these formulations will increase in the foreseeable future. Generic SIR products are not available in the EU and Australia, which is most likely caused by the limitations associated with patent terms. As mentioned above, there have recently been 8 available generic Rapamune, but there are no reports of bioequivalence studies in the literature. Nevertheless, Bolar exemption (known in the United States as Roche-Bolar exemption) allows the bioequivalence of the original drug to be tested and to prepare its generic before the expiry of the patent period. A more generic SIR is expected to appear immediately after the end of all the protective patent terms and exclusive rights for the original Rapamune (Fig. 5).
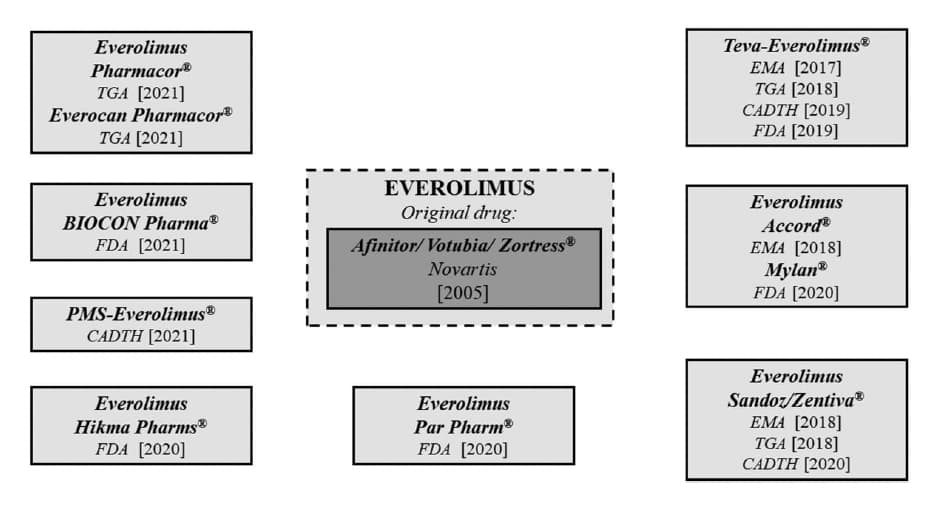
EVE (Everolimus)
EVE was introduced in 2005 by Novartis under the Afinitor/Votubia/Certican names in the EU, and in 2009 in the United States and Canada. It is available in different strengths (0.25 mg—0.5 mg—0.75 mg—1 mg) as oral tablets and tablet for suspension. The first generic was preliminarily introduced by Teva Europe. Subsequently, in 2018, this manufacturer introduced generic EVE under the same brand name in Australia, and in 2019 in the United States and Canada. The 2 next generic forms of EVE were introduced in 2018 by Accord and Sandoz/Zentiva in the EU drug market. The last was available in Australia and Canada in 2018 and 2020, respectively. In the past 2 years, 3 generics were initially approved in the United States by the Everolimus Hikma Pharms, Everolimus Par Pharm in 2020, and Everolimus BIOCON in 2021. Everolimus-PMS was introduced in Canada at the beginning of 2021.
As in the case of SIR, it is expected that a more generic EVE will appear immediately after the end of all protective patent terms for the original form (Fig. 6). A few patent terms and exclusive rights expired in the United States in 2020 (and previously in the EU), which allows new EVE generics to be introduced in the drug market.
Generic_Medicinal_Products_in_Immunosuppressive.8.pdf (740.4 KB)