This podcast series covers longevity biotech and longevity science:
NFX General Partner Dr. Omri Drory on how to end involuntary death
Full transcript here: NFX General Partner Dr. Omri Drory on how to end involuntary death
Gemini Pro AI Video Summary and Analysis:
This analysis examines the interview with Dr. Omri Amirav-Drury, General Partner at NFX, regarding the economic, biological, and philosophical imperatives for radical life extension.
A. Executive Summary
Dr. Omri Amirav-Drury posits that aging is the primary root cause of 90% of deaths in the Western world and represents the most significant market opportunity in human history. The core thesis of the discussion is that human lifespan is poised for a third “doubling”—moving from the current median of 80 to 160 years—and that once this threshold is crossed, the “longevity escape velocity” will effectively render death optional.
Drury argues that longevity is the singular technological solution to America’s most pressing economic crises: the exploding national debt, the insolvency of Social Security, and declining fertility rates. By extending the “peak earning years” and maintaining biological youth (staying “22 as long as you want”), individuals could self-retire, interest rates would drop due to capital accumulation, and the GDP would grow through sustained productivity. He dismisses concerns of overpopulation, noting that the real existential threat is the exponential decline in global fertility (e.g., South Korea’s demographic collapse).
The “ABCD Plan for Aging” is introduced as a strategic framework:
- Plan D (Death): The current, suboptimal default.
- Plan C (Cryonics): “Stopping time” as an insurance policy.
- Plan B (Bioengineering): Solving diseases one by one (limited by the “whack-a-mole” nature of aging).
- Plan A (Alternative Bodies/Replacement): Utilizing synthetic embryos (stroids) to grow genetically identical, young organs and tissues for replacement.
Drury is critical of the “healthspan” narrative, calling it a “chicken” approach to avoid the political friction of discussing immortality. He emphasizes that AI, while powerful for protein folding and genomic analysis, is not a “magic wand” because biology requires empirical “wet lab” validation which cannot be bypassed by pure computation. Ultimately, he defines the current era as a “Race to 160,” where the primary bottlenecks are no longer just scientific, but cultural and ethical acceptance of technologies like synthetic embryology and germline optimization.
B. Bullet Summary
- The Doubling Hypothesis: Human lifespan doubled from 20 to 40, then 40 to 80; the next doubling to 160 is the final “solved” state.
- Economic Solvency: Radical longevity eliminates the need for Medicare/Medicaid and makes Social Security sustainable by extending productive work years.
- Fertility Crisis: Longevity is the only viable solution to sub-replacement fertility rates in developed nations like Japan and South Korea.
- Plan A Superiority: Nature already knows how to reset biological age (embryogenesis); we should use stem-cell-derived embryos to grow replacement parts.
- The “Stroid” Unlock: Creating human embryo models from stem cells (without sperm or egg) is the key to infinite young tissue.
- AI vs. Biology: AI lacks data and simulation accuracy; “wet lab” experiments are still the rate-limiting step in biotech.
- Longevity Escape Velocity: Once we solve current late-life diseases to reach 160, no “new” diseases are expected to emerge at age 400.
- Optics of Wealth: Early expensive treatments are necessary; eventually, they scale and become cheaper than the cost of elderly care.
- Biological “Code”: Even simple organisms like E. coli function on complex “if-then” logic gates encoded in DNA.
- The “Healthspan” Critique: Focusing only on healthspan is a defensive, PC maneuver; the goal should be “ending involuntary death.”
- VC vs. Sugar Daddies: Radical longevity often requires “Sugar Daddies” (Ultra-high-net-worth individuals) because the 10-year VC fund cycle is often too short for biological breakthroughs.
- Synthetic Embryo Ethics: Drury supports using stem-cell embryos for therapy but draws a hard ethical line at growing full humans for parts.
- Cultural Shift: In the future, “old-fashioned” unoptimized procreation may be viewed as irresponsible or “crazy.”
- The “Tech Tree”: Just as GPUs unlocked AI, synthetic embryology and CRISPR are unlocking the next phase of human evolution.
- Longevity as Moral Imperative: It is “immoral” not to work on ending aging given the scale of human suffering it causes.
D. Claims & Evidence Table (Adversarial Peer Review)
| Claim from Video | Speaker’s Evidence | Scientific Reality (Best Available Data) | Evidence Grade (A-E) | Verdict |
|---|---|---|---|---|
| Lifespan Doubling to 160 | Historical trend (20 to 40 to 80). | The maximum documented human lifespan is 122 (Jeanne Calment). Biological “hard limits” (Gompertz Law) suggest 120-150 is the ceiling without radical intervention. | E (Expert Opinion) | Speculative |
| Synthetic Embryos for Parts | Renewal Bio’s work on “stroids” at Weizmann Institute. | Stem-cell-derived embryo models have reached 14-day equivalents in vitro. Scaling to organogenesis for transplant is unproven in humans. | D (Pre-clinical/In vitro) | Plausible (Emerging) |
| Partial Reprogramming | Mentions Yamanaka factors (OSKM) and companies like Retro/Altos. | In vivo reprogramming has rejuvenated tissues in mice. Human safety is unknown; risk of teratomas (tumors) is high. | D (Animal Models) | Translational Gap |
| Economic Fix via Longevity | Personal analysis of GDP (Population x Productivity). | The “Longevity Dividend” paper suggests a 1-year increase in life expectancy is worth $38 trillion to the US. Logic is sound but relies on “healthspan” success. | C (Economic Modeling) | Plausible |
| Aging as Main Risk for Smoking | Comparison of bar charts for lung cancer risk factors. | SEER data confirms age is the single greatest risk factor for most cancers, though smoking is the primary modifiable risk for lung-specific cancer. | C (Cohort/Epidemiological) | Strong Support |
| GLP-1s for Alzheimer’s | Mentions obesity drugs affecting multiple indications. | Ongoing Phase 3 trials (EVOKE) are testing Semaglutide for early Alzheimer’s. Mechanisms (neuroinflammation) are plausible but results are pending. | B (Ongoing RCTs) | Plausible |
E. Actionable Insights
Top Tier (High Confidence)
- Biological Basics: Adhere to the “80/20” of health: weight management, sleep hygiene, and community engagement. These are the only current interventions with “Level A” support for reaching the current 80-120 year limit.
- GLP-1 Awareness: Monitor the evolution of GLP-1 agonists (Semaglutide/Tirzepatide) not just for weight loss, but for systemic anti-inflammatory benefits that may impact neurodegeneration.
Experimental (Risk/Reward)
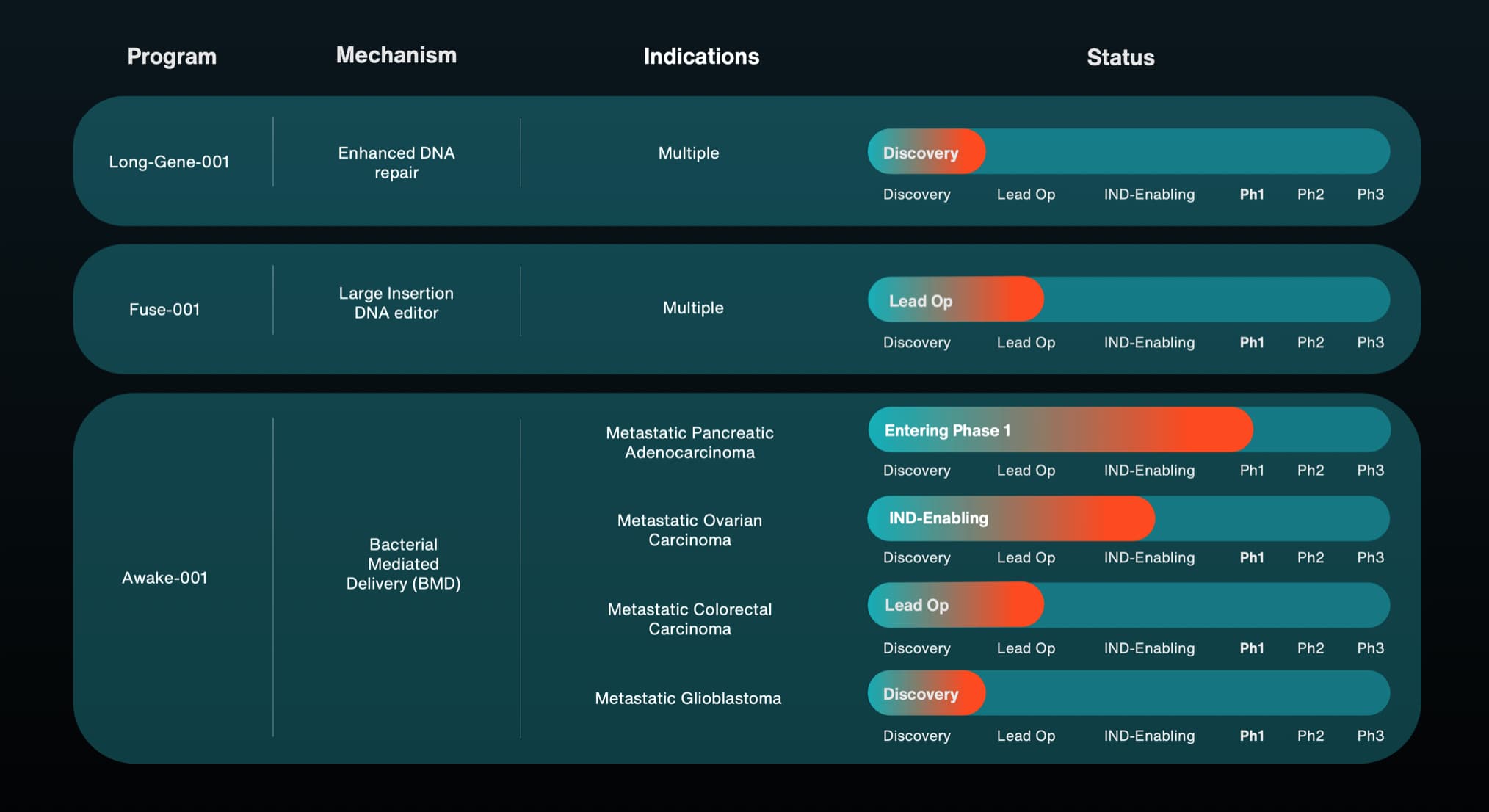
- Synthetic Embryology Monitoring: Track the progress of Renewal Bio and similar entities. While not currently actionable for consumers, this represents the “Plan A” for organ replacement within the next 20 years.
- Dog Longevity: For pet owners, monitor Loyal (Cellular Longevity, Inc.). Their progress with the FDA on aging as a primary endpoint will set the regulatory precedent for human drugs.
Avoid
- Supplement Hype: Drury notes a lack of FDA-approved drugs that “move the needle.” Avoid high-cost “longevity” supplements that lack human RCT data for lifespan extension.
- Wait-and-See (Plan D): Drury explicitly warns against waiting to “get rich” before caring about longevity. The biological decay (Plan D) is happening now.
H. Technical Deep-Dive: The “Stroid” Mechanism
The core technology discussed is Stem-cell-derived Embryo Models (Stroids). Unlike traditional cloning (Somatic Cell Nuclear Transfer), this involves:
- Pluripotency Induction: Reverting adult skin cells to a naive pluripotent state.
- Self-Organization: Under specific biochemical cues, these cells organize into structures mimicking post-implantation embryos without the need for fertilization.
- Ex-utero Growth: Using specialized bioreactors (like the one developed by Jacob Hanna), these models can be grown to stages where organ primordia (heart, brain, gut) appear.
- The “Tech Tree” Goal: The objective is to harvest these “zero-year-old” tissues for autologous (genetically identical) transplants, bypassing the MHC-mismatch issues seen in traditional organ donation.
I. Fact-Check
-
Claim: “Nine of the 10 things that kill us… the root cause is aging.”
-
Verification: According to the CDC, heart disease, cancer, COVID-19 (in 2021-23), stroke, and Alzheimer’s are top killers. All show exponential incidence increases with age. Verified.
-
Claim: “America spends more on interest payments than the army.”
-
Verification: As of late 2024/2025 CBO reports, net interest costs on the national debt have indeed surpassed the Department of Defense budget. Verified.
-
Claim: “South Korea has one child per woman.”
-
Verification: The World Bank and local reports indicate the rate has actually dropped below 1.0 (approx 0.72 in 2023). Verified (Actually worse than stated).