i LOVE guanfacine because it can increase mental capacity and output while being the EXACT opposite of a stimulant
instead of making the brain a clusterfuck it decreases firing making you calm and clear headed instead of all over the place
- calm focus
- better working memory
- “zen” flow state
- better mental resilience to stress
- no hyperactivity
- less distraction
how the fuck does it do that since it literally decreases neural signaling?
guanfacine is a selective agonist of the α2A receptor, a receptor located primarily in the prefrontal cortex which could be named the “control conter of mental performance”.
activation of α2A by guanfacine results in less intracellular cascades going on that would normally make you stimulated.
it makes your brain less noisy so that you can think. clearly.
now you need to get the dosing right because it can be sedative at higher doses. anywhere in between 1-2mg is enough.
I’ve heard good things about it. I’m about to trial some ADHD medication and this is something I’m interested in adding in conjunction with a stimulant.
selective agonist of the α2A receptor
Sounds good! Is there safety/longevity data on this?
It’s approved for human use so it is somewhat safe.
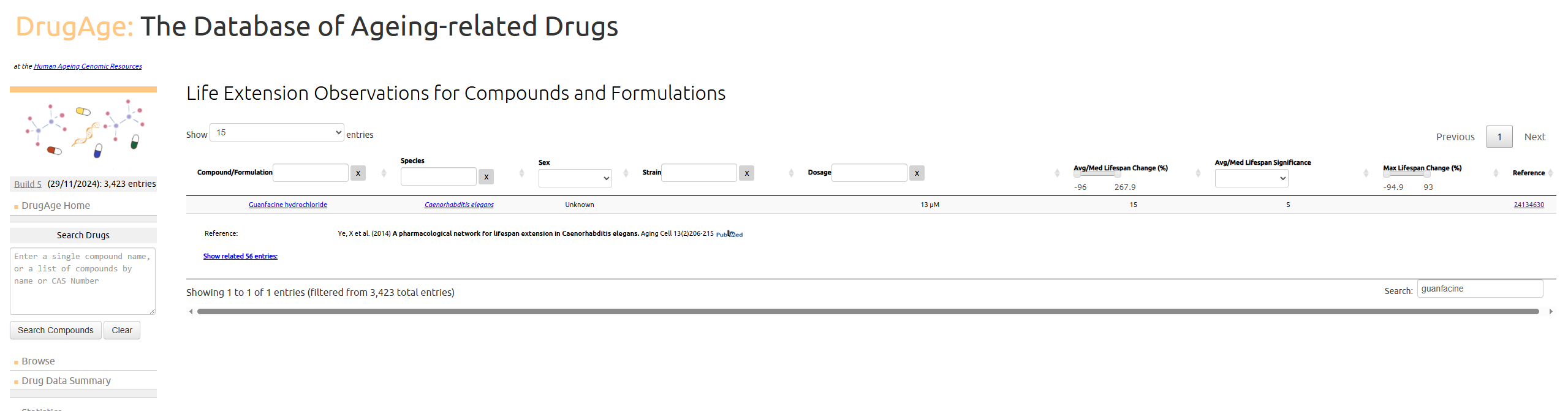
As for longevity I did find one study that mentioned it for worm lifespan effects, according to DrugAge it increased worm lifespan by 15%
But actually reading the study I struggle to find where it specifically mentions what guanfacine does: https://onlinelibrary.wiley.com/doi/10.1111/acel.12163
Mechanism and Selectivity
These agents function by stimulating alpha-2 receptors in the central nervous system, which inhibits the firing of sympathetic nerves.
Guanfacine: Selective for the alpha-2a subtype. This selectivity is associated with fewer sedative side effects compared to non-selective agents.
Clonidine: Non-selective (binds alpha-2a, 2b, and 2c). While effective at reducing sympathetic tone, it is associated with higher rates of sedation and hypotension.
Clinical Efficacy in Combination
The literature provides specific evidence for the utility of combining alpha-2 agonists with methylphenidate to manage cardiovascular side effects. A longitudinal case study of a patient with renal disease (ADPKD) and ADHD reported that monotherapy with methylphenidate (18 mg) caused hypertension and tachycardia, while monotherapy with guanfacine caused hypotension. The combination of Methylphenidate (18 mg) and Guanfacine (4 mg) successfully stabilized blood pressure (<110/75 mmHg) and managed symptoms, effectively balancing the “push” of the stimulant with the “pull” of the alpha-agonist.
This combination strategy addresses the “decrease in cardiac baroreflex sensitivity” observed with methylphenidate use, potentially restoring autonomic responsiveness.
https://aristotle.science/share/thread/thr_vsWmagdxt657q7cxVeifvfEB
5.4. Guanfacine protects PFC neurons from psychological and physiological stress
Evidence from both in vivo and in vitro studies show that guanfacine can protect PFC circuits from stress exposure, including preventing atrophy due to chronic stress exposure. This has been seen for both psychological stressors such as restraint stress in rodents (Hains et al., 2015), as well as for physiological stressors such as reduced oxygen (Kauser et al., 2013). As both psychological and physiological stressors drive feedforward calcium-cAMP-PKA-K+ signaling and impair working memory, guanfacine’s ability to inhibit these actions may preserve network connectivity and PFC function (schematically shown in Fig. 6A).
Fig. 6. Guanfacine counteracts the effects of chronic stress in PFC. (A) A schematic illustration showing how stress increases, while guanfacine inhibits, the feedforward Ca2±cAMP-PKA-K+ actions that weaken PFC network connectivity, neuronal firing and function. α2A-AR are also expressed on activated microglia, where α2A-AR stimulation deactivates microglia and thus has anti-inflammatory actions. (B) Example of a layer II/III mPFC pyramidal cell distal dendrite from a control vs. chronic stressed rat, showing the reduced spine density in the stressed PFC. Scale bar indicates 25 μm. Adapted from (Hains et al., 2009) with permission. (C) Chronic stress (Str) exposure reduces PFC spine density in vehicle-treated rats, but not in those receiving daily guanfacine treatment. Con = control; Str = chronic stress; Veh = vehicle; Gfc = guanfacine. Adapted from (Hains et al., 2015) with permission.
In addition to direct actions on dendritic spines, guanfacine’s protective effects may involve anti-inflammatory actions through α2A-AR on glia (Gyoneva & Traynelis, 2013). As schematized in Fig. 6A, α2A-AR are expressed on activated microglia, where their engagement deactivates microglial activity (Gyoneva & Traynelis, 2013). Thus, guanfacine may reduce microglial phagocytosis of spines. The anti-inflammatory effects of α2A-AR agonists are well-established, and contribute to their widespread use as co-anesthetics in surgery, where they reduce the incidence of emergence delirium (Zhang et al., 2020). These anti-inflammatory effects may also contribute to guanfacine’s protections from hypoxia in animal models (Kauser et al., 2013, Kauser et al., 2016), and from encephalitis in humans (Singh-Curry, Malhotra, Farmer, & Husain, 2011). Thus, guanfacine’s engagement of α2A-AR in PFC may have coordinated actions, both strengthening connections on spines and reducing local phagocytic actions by glia.
My MD has me using guanfacine along with ritalin. It enables a lower dose of Ritalin and it also helps my sleep architecture as measured by an EEG device. I am a huge fan.
I agree with your description of how it helps.
I just got 30-day supply from asking PCP
they recommend you take it at night