Paper:
The Vascular Senohub: How Aging Arteries Orchestrate Systemic Decline
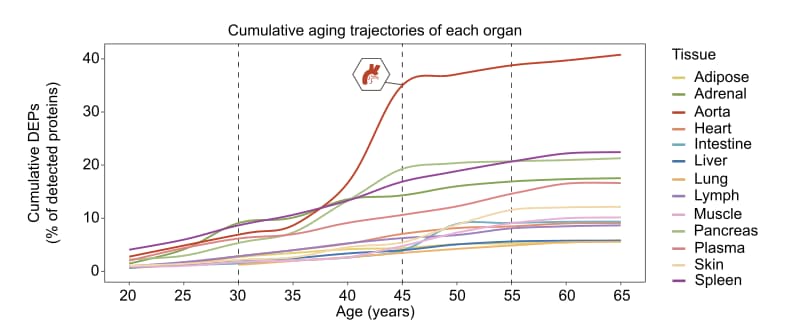
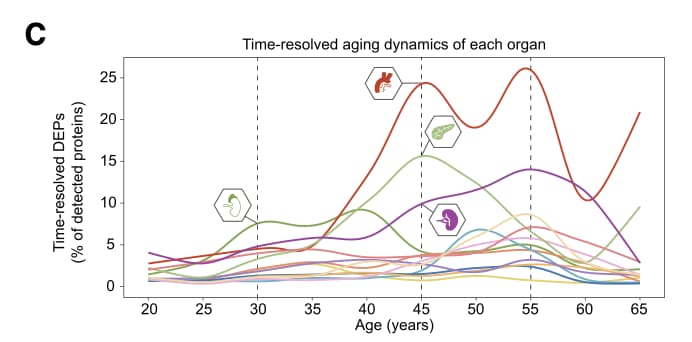
A groundbreaking 50-year human proteome atlas has mapped the trajectory of human aging across 13 organs, revealing that our blood vessels act as early instigators of systemic decline. While chronological age advances at a constant speed, biological aging occurs in asynchronous leaps, with the aorta experiencing a massive proteomic inflection around the age of 50.
By analyzing 516 tissue samples alongside matched transcriptomics, researchers uncovered a widespread decoupling between mRNA and protein levels in older tissues. This proteostasis collapse is driven by a failure in translation machinery and chaperone networks, culminating in the toxic accumulation of amyloid proteins (such as SAP and SAA1/2) across organs. This localized amyloid buildup triggers an inflammatory cascade involving immunoglobulins and the complement system.
The central discovery is the role of the vasculature as a “senohub.” The aorta is not merely passively damaged over time; it actively accelerates body-wide aging. By profiling the senescence-associated secretory phenotype (SASP) across tissues, the team identified specific circulating “senoproteins”—most notably GAS6 and GPNMB—that are overproduced by aging blood vessels and secreted into the bloodstream. When these proteins are applied to healthy cells or injected into young mice, they act as gerontogens, inducing DNA damage, cellular senescence, and physical frailty. GAS6, for example, binds to TAM receptors to drive endothelial dysfunction, transferring the aging phenotype from the vascular wall to peripheral organs like the liver and spleen.
This multi-organ proteomic clock shifts the paradigm from focusing solely on localized tissue damage to understanding aging as a contagious, systemic network driven by vascular-derived factors.
Institution: Chinese Academy of Sciences (Institute of Zoology, Beijing Institute of Genomics) & West China Hospital of Sichuan University Country: China Journal: Cell
The impact score of this journal is 45.5, evaluated against a typical high-end range of 0–60+ for top general science, therefore this is an Elite impact journal.
Mechanistic Deep Dive
- Mitochondrial Dynamics: Aged tissues demonstrated a ubiquitous downregulation of core mitochondrial proteins essential for biogenesis, translation, and respiratory electron transport, directly contributing to organ-specific metabolic decline. [Confidence: High]
- Proteostasis & Autophagy: The study identifies a severe divergence between the transcriptome and proteome in aged tissues, driven by the downregulation of TRiC/CCT chaperonins, proteasomal subunits, and chaperone-mediated autophagy networks. This results in the pathogenic accumulation of amyloid proteins (SAP, SAA).
- Inflammation & Viral Mimicry: Aging tissues show upregulated MAVS-TRADD complexes and reactivation of endogenous retroviruses (MMTV), leading to profound inflammasome activation and SASP cytokine secretion. [Confidence: Medium]
- Organ-Specific Priorities: The aorta undergoes the earliest and most severe proteomic remodeling (peaking between ages 45–55). It operates as a primary senohub, selectively enriching circulating gerontogens like GAS6. GAS6 binds to TAM receptors (TYRO3, AXL, MERTK) to induce systemic aging cascades in distant tissues. [Confidence: High]
Novelty
Prior proteomic clocks relied heavily on plasma analysis without identifying the exact tissue of origin for circulating biomarkers. This study physically maps the proteomic aging network across 13 solid organs and pairs it with plasma data, pinpointing exactly where circulating aging biomarkers originate. It establishes the aorta not just as a target of cardiovascular aging, but as an active endocrine driver of systemic decline via the secretion of GAS6 and GPNMB.
Critical Limitations
- Translational Uncertainty: Human samples were derived from victims of severe traumatic brain injury. The systemic shock and inflammatory storm preceding postmortem organ procurement almost certainly altered the proteomic landscapes, injecting confounding variables into the baseline “healthy” aging signature. [Confidence: High]
- Methodological Weaknesses (In vivo): The mouse validation relies on an acute dosing protocol (4 intravenous injections over 20 days) in relatively young mice (8 months). This model mimics an acute inflammatory or toxic insult rather than the chronic, low-grade accumulation of senoproteins seen in natural biological aging.
- Missing Data: Critical longevity-modulating organs (the brain, kidneys, and reproductive system) are entirely absent from the atlas. Furthermore, there is no longitudinal survival data to prove that inhibiting circulating GAS6 or GPNMB extends maximum or median lifespan. Future interventions targeting these pathways will require robust lifespan assays to verify true geroprotective efficacy.