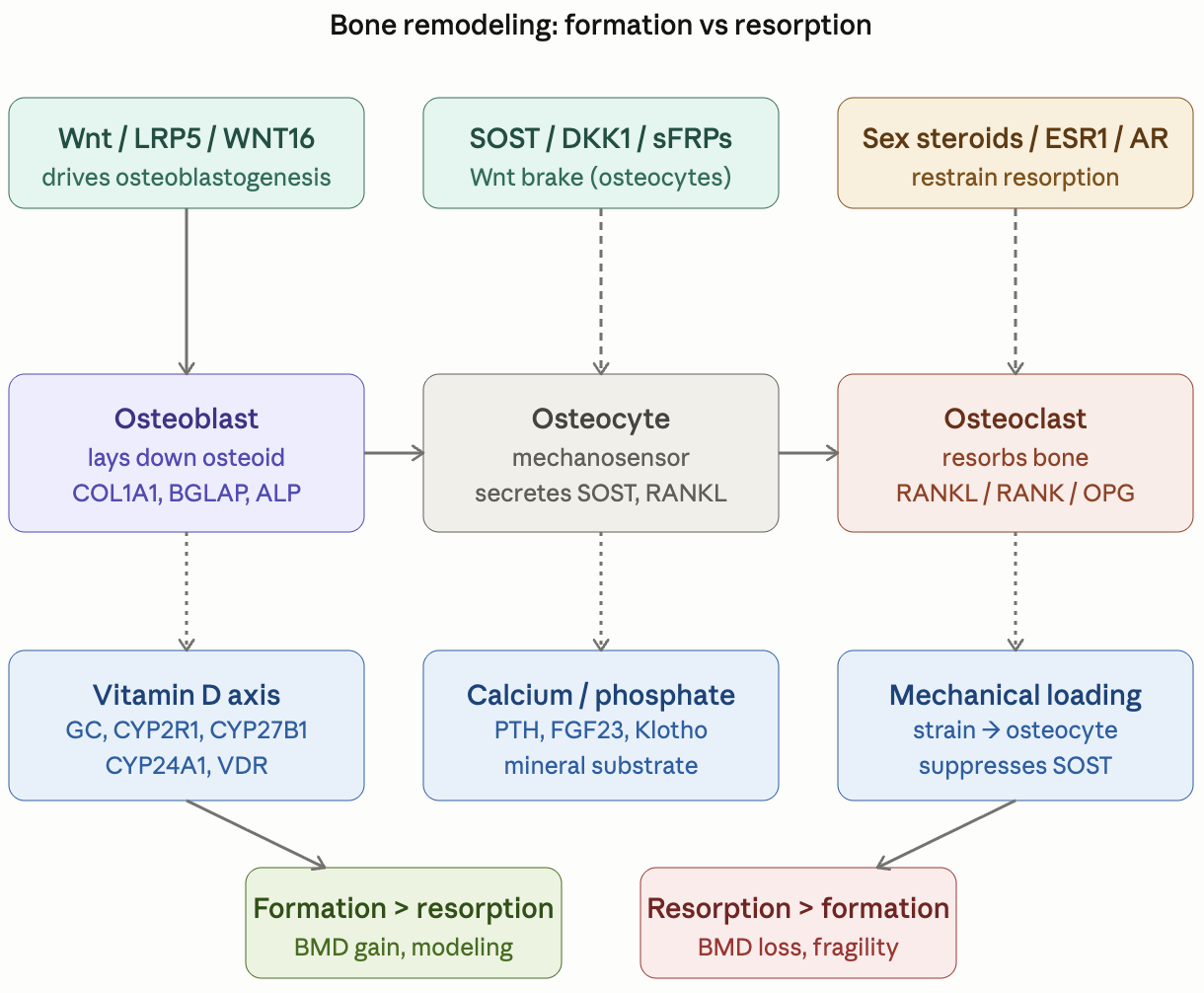
I’m continuing my deep dives into the genetic pathways to get actionable insights as the previous ones have been incredible precise and useful. This time I’m looking at the bone remodeling related genetic pathways.
Here is the general description of the pathways and their variants. I will put the finding about my own genome below it as an example of what useful and actionable insights you can get.
Bone_Skeletal_Aging_Genetic_Pathway_Reference.pdf (589.9 KB)
The pdf report above is valid for everybody but here is the summary of the findings I get when I apply it to my own genome.
Top findings (highest evidence + highest expected impact)
| # | Finding | Evidence | Why it matters for this person |
|---|---|---|---|
| 1 | OPG (TNFRSF11B) homozygous low-OPG haplotype — three OPG SNPs hom-variant: rs2073618 K3N C/C (DP=51, GQ=99), rs6469804 A/A (DP=118), rs6993813 C/C (DP=80); plus rs6469792 het | A | The single strongest convergence in the profile. K3N reduces OPG signal-peptide cleavage and lowers serum OPG (Hsu 2006 JBMR; Guo 2010 meta-analysis); intronic hom-variants further reduce OPG transcription. Net: genetically elevated baseline RANKL:OPG ratio — the molecular target of denosumab. |
| 2 | RANKL + RANK heterozygous risk burden — TNFSF11 rs9594738 + rs9594759 het; TNFRSF11A rs884205 het | A | Compounds finding 1. Both RANKL GWAS SNPs are Estrada 2012 P<10⁻¹⁰ BMD loci. Combined with hom OPG, every node of the resorption-drive axis points the same direction. |
| 3 | VDR signaling-block hom haplotype — FokI rs2228570 f/f, BsmI rs1544410 T/T (BAM-confirmed 89/89 reads), ApaI rs7975232 A/A, TaqI rs731236 G/G; Cdx2 absent | B | Substrate-saturated, receptor-limited configuration. Current 25(OH)D 84.6 ng/mL (Labcorp 01/07/2026) is at/above VDR saturation; further substrate provides no marginal benefit. Reduces intestinal Ca absorption efficiency per unit 1,25(OH)₂D. |
| 4 | WNT16 / CPED1 BMD-locus heterozygous burden — rs3801387 (lead BMD SNP, P=2.6×10⁻³¹), rs917727 (perfect LD), rs2707466 (Thr→Ile, CBT P=6.2×10⁻⁹), rs2908004 (Gly→Arg, BMD P=1.2×10⁻¹⁵) | A | Het at four GWAS-replicated SNPs at the strongest BMD-Wnt locus. WNT16 specifically induces osteoblast OPG production — reduced WNT16 signaling AMPLIFIES the OPG deficit (findings 1+4 are not independent). Cumulative ~0.10–0.15 SD lower BMD. |
| 5 | SOST elevated genetic floor — 8 of 9 SOST variants het, including BOTH Van Buchem-region SNPs (rs7220711, rs1107748) with direct ChIP/reporter mechanistic evidence (Maupin 2019 Bone) | B | Sclerostin is the dominant Wnt brake responding to mechanical loading. Higher genetic floor means loading must overcome more brake to drive osteoblast formation. The genetic profile mimics, in heterozygous form, the converse of the Van Buchem high-bone-mass deletion — making romosozumab uniquely genotype-aligned IF ever clinically indicated. |
| 6 | COL1A1 + matrix-protein burden — COL1A1 Sp1 het (Ss, the highest-injury-risk genotype in Mann 2001 J Clin Invest) PLUS COL1A1 −1997 promoter hom-variant; COL1A2 het, BGLAP het, SPP1 het, SPTBN1-region hom | B | Matrix-protein transcription/post-translation non-default at multiple sites. Sp1 het = 1.4× fracture OR INDEPENDENT of BMD (Mann 2001 meta-analysis) — fracture-resistance per unit BMD is reduced. The Strength report framed COL1A1 Sp1 as tendon risk only; bone-context expansion is formalized in this report. |
| 7 | CYP2R1 dual heterozygous rs10741657 + rs12794714 — ~0.10 SD reduced 25(OH)D production efficiency (Jiang 2018 Nat Commun, n=417,580) | A | Already documented in Glucose / Strength reports. Compounded with intact substrate transport (GC favorable, see #10) and intact catabolism (CYP24A1 favorable, see #10), this explains the substrate-saturated state on D3 5000 IU EOD. |
| 8 | AXIN1 rs9921222 hom T/T — Wnt destruction complex variant; OR=16.6 vs C/C in Egyptian case-control (Nassar 2023 BMC Musculoskelet Disord, n=101) | C | Largest single nominal effect size in the profile but smallest evidence base — no European-ancestry replication; CI very wide (2.0–136.4). Conservatively flag as moderate-effect not high-effect. Mechanistically aligned with the Wnt-brake-and-antagonist stack. |
| 9 | ESR1 PvuII het + XbaI absent → protective haplotype not constructible | A | Genomos consortium n=18,917 (Ioannidis JAMA 2004): the PX/PX haplotype (PvuII-C + XbaI-G) carries ~30% lower fracture risk. With XbaI rs9340799 hom-reference (BAM-confirmed 69/69 reads as A), this person CANNOT have PX/PX. Net status = neutral, not protective. |
| 10 | Genetically protected: GC favorable + CYP24A1 favorable + LRP5 LoF burden absent + Klotho/CASR/PTH/FGF23/TRPV6 reference | A | The strongest 25(OH)D-lowering SNP (GC rs2282679) is absent; Gc2-defining rs4588 absent. CYP24A1 rs6013897 + rs17216707 absent. LRP5 rs3736228 hom-reference (BAM-confirmed 79/79 reads as C). Mineral-homeostasis machinery genetically intact across 6 panel SNPs. |