This was launched the other week, hopefully a valuable step forward in nailing down a suite of generally agreed-upon and validated biomarkets for aging. Video overview of their plans here:
More information:
Papers they have “in process”
Timeline: September 2022 - February 2023 (manuscript currently under review)
Title: Biomarkers of Aging for Identification and Evaluation of Longevity Interventions
Authors: Biomarkers of Aging Consortium; Mahdi Moqri; Chiara Herzog; Jesse Poganik; Jamie Justice; Daniel Belsky; Albert Higgins-Chen; Alexey Moskalev; Georg Fuellen; Alan Cohen; Ivan Bautmans; Martin Widschwendter; Jingzhong Ding; Alexander Fleming; Joan Mannick; Jing-Dong Jackie Han; Alex Zhavoronkov; Nir Barzilai; Matt Kaeberlein; Steven Cummings; Brian Kennedy; Luigi Ferrucci; Steve Horvath; Eric Verdin; Andrea Maier; Michael Snyder; Vittorio Sebastiano; Vadim Gladyshev
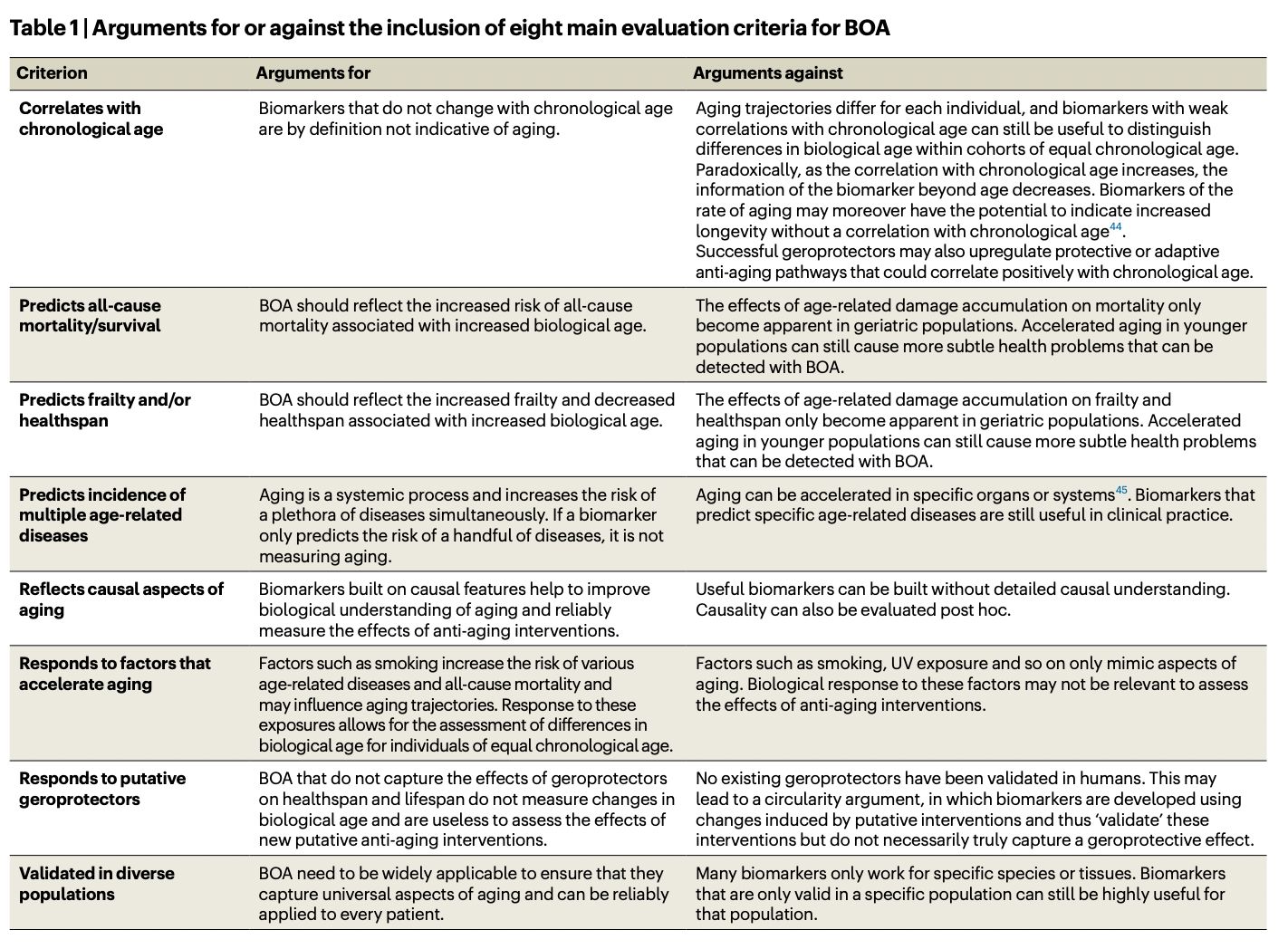
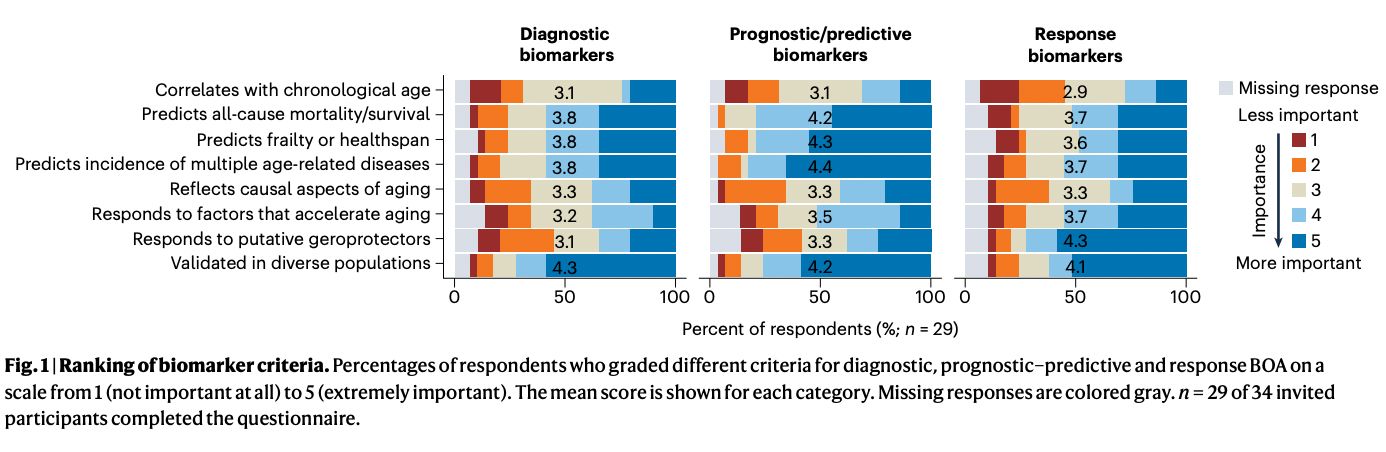
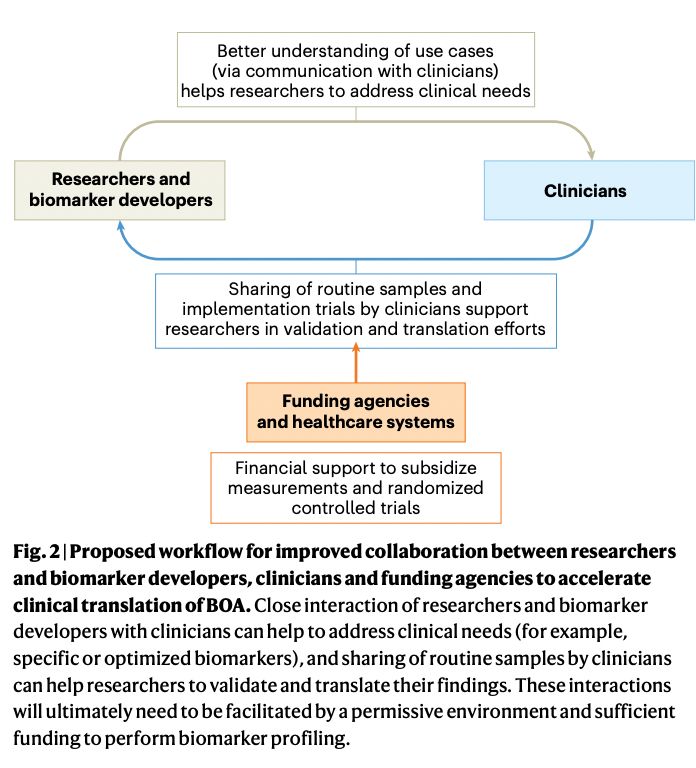
Abstract: With the rapid expansion of aging biology research, identification and evaluation of longevity interventions in humans have become key goals of this field. Biomarkers of aging are critically important tools in achieving these objectives over realistic timeframes. Here, we advance a framework for characterizing and assessing biomarkers of aging, including consensus terminology for several key concepts in this space. We propose classification of existing biomarkers and evaluate their feasibility, age-sensitivity, mechanistic underpinnings, and response criteria. We also discuss analytical and clinical validation of biomarkers of aging, including validation as surrogate endpoint biomarkers of clinical outcomes. This framework sets the stage for the future development of valid biomarkers of aging and their ultimate utilization in clinical trials and practice.
Timeline: March 2023 - Dec 2023 (manuscript currently being drafted)
Title: A framework for evaluation of blood-based biomarkers of aging
Authors: Biomarkers of Aging Consortium; We are inviting additional experts to work on validation of biomarkers of aging. Interested in joining this workgroup?
Abstract: Blood-based biomarkers that quantify biological aging have recently gained popularity due to their ability to predict aging-related outcomes and their potential to evaluate the efficacy of pro-longevity interventions. However, there is a clear need for a consensus framework for systematic validation of biomarkers of aging to enable their advancement to the clinic. Here, we review current validation efforts for predictive blood-based composite biomarkers of aging in population studies, discuss the challenges in compatibility and generalizability of these efforts, and provide recommendations to facilitate future validation of biomarkers of aging. Finally, we discuss how systematic validation of predictive biomarkers of aging can accelerate biomarker development and ultimate application in clinical trials.