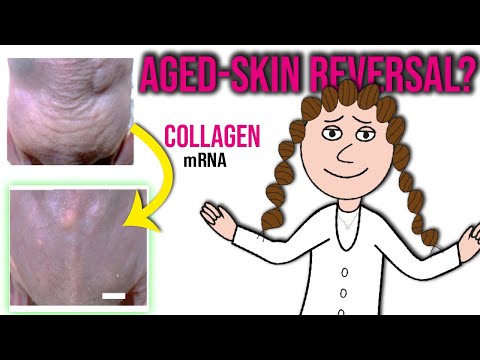
A skin patch with microneedles that inject mRNA reduced wrinkles in mice with UV-damaged skin, a technique that could be used to treat skin conditions and arthritis related to collagen deficiency
Using mRNA to direct the body to make more collagen – the most abundant protein in skin – reduces wrinkles in mice. This therapy could be useful to treat other disorders related to collagen loss, including some genetic skin conditions and arthritis.
Wrinkles form, in part, because UV radiation from the sun damages collagen fibres in skin, causing it to lose structure and elasticity. Medical treatments that use mRNA, such as covid-19 vaccines, provide cells in the body with genetic instructions for producing proteins. To test whether this could be used to replace destroyed collagen, Betty Kim at the MD Anderson Cancer Center in Texas and her colleagues injected mRNA into hairless mice that had been exposed to UV radiation for 60 days to trigger wrinkles.
The researchers packaged the genetic recipe for collagen in tiny molecular messenger bags called extracellular vesicles. They then injected four of the mice with five doses of these extracellular vesicles, while another four mice received topical retinoid treatment – a common medication for skin ageing. A separate quartet of mice didn’t receive treatment or UV exposure.
Kim and her team used microscope imaging to track the number of wrinkles the mice developed. After 28 days, those injected with the mRNA had the same number of wrinkles, on average, as mice that were not exposed to UV light, and half the number of wrinkles as mice treated with retinoids.
This effect slowly wore off over an additional four weeks, with wrinkles returning to pre-treatment levels after 56 days. Though, a subsequent experiment found that delivering the mRNA via a skin patch with microscopic needles reduced wrinkles in mice for about twice as long, on average, as injections.
Nicholas Gulati at Mount Sinai Hospital in New York says this is the first dermatological mRNA treatment developed, and a novel approach to delivering mRNA medicine. Currently, mRNA is packaged inside carriers called lipid nanoparticles, which can trigger an overreactive immune response, inciting inflammation and even anaphylaxis. Extracellular vesicles don’t induce this reaction, as they naturally occur in the body. In fact, mice treated with the collagen-replacing mRNA exhibited no redness or swelling.
“The application for this type of technology is limitless,” says Kim. “Not only can it be used for skincare, but also to potentially treat genetic disorder and even cancers.”
For instance, these mRNA injections could treat osteoarthritis, which occurs when the collagen-rich cartilage that cushions joints deteriorates. She says the technology could also be used with different types of mRNA to treat brain cancer, as extracellular vesicles can bypass the blood-brain barrier, which prevents pathogens and many medications from reaching the brain.
Journal reference: Nature Biomedical Engineering, DOI: 10.1038/s41551-022-00989-w
Intradermally delivered mRNA-encapsulating extracellular vesicles for collagen-replacement therapy
The success of messenger RNA therapeutics largely depends on the availability of delivery systems that enable the safe, effective and stable translation of genetic material into functional proteins. Here we show that extracellular vesicles (EVs) produced via cellular nanoporation from human dermal fibroblasts, and encapsulating mRNA encoding for extracellular-matrix α1 type-I collagen (COL1A1) induced the formation of collagen-protein grafts and reduced wrinkle formation in the collagen-depleted dermal tissue of mice with photoaged skin. We also show that the intradermal delivery of the mRNA-loaded EVs via a microneedle array led to the prolonged and more uniform synthesis and replacement of collagen in the dermis of the animals. The intradermal delivery of EV-based COL1A1 mRNA may make for an effective protein-replacement therapy for the treatment of photoaged skin