chatGPT:
Below is a tidied version of the transcript, followed by a summary and critique.
Tidy transcript
Host:
Hey everyone. When it comes to longevity, a big focus of this channel is slowing the rate of aging using a testing-, tracking-, and intervention-based approach. But this approach only goes so far. What about rejuvenation?
Today, Dr. Michael Levin and Leo Lopez will present evidence for restoring bioelectric gradients as a potential top contender—or what I see as a potential top contender—for treating aging, cancer, and human rejuvenation.
So welcome, Michael and Leo. Let’s get into the presentation.
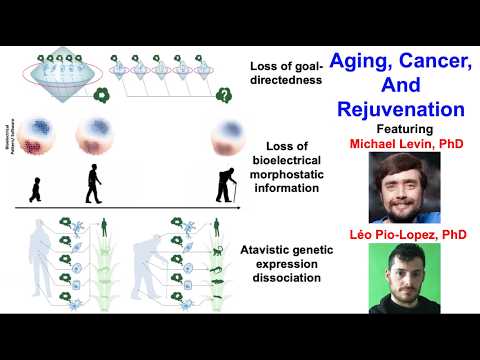
Michael Levin:
Excellent. Thanks for having us. I’m Mike Levin, and this is Leo Pio Lopez, a senior scientist in my group.
What we want to do today is talk about three specific things, one of which is bioelectricity. There are three fundamental theories of aging concerning the ultimate root cause.
First, there are damage theories, which say aging is basically the accumulation of thermodynamic noise and damage, so aging is fundamentally a problem of physics.
Second, there are programmatic theories, which suggest evolution has favored a phenotype of decline during aging, so aging is due to a problem of biology or evolution.
And we’re going to talk a little bit about a third kind of theory, basically a cognitive theory, which suggests that aging is, at least in important part, a problem of cognition or cybernetics. It’s a fundamental problem that doesn’t reduce simply to physics or biology, although of course these are not mutually exclusive.
In our lab we have three current contributions to the aging field. My group doesn’t primarily work on aging, but we have three different things we’re working on and we’re going to show you all three of those.
One is this notion of a loss of goal-directedness as a fundamental cause of aging. Then we’re going to talk about the degradation of instructive patterns that happen to be bioelectric, and aging as a consequence of the degradation of this instructive information. And then something fairly amazing that Leo discovered called atomistic dissociation, which is one of the consequences of aging.
If aging is going to be a widespread, body-system-wide reduction of function, we need to ask what is upstream. What is actually being disrupted to cause all this?
One of the things we’ve proposed is that aging is a loss of morphostatic information—a loss of information that normally keeps the cells in the body orchestrated toward a healthy, functional structure.
Specifically, I want to introduce the idea of bioelectricity. It’s long been known that bioelectricity is important in the brain. It’s the thing that makes you more than a pile of neurons. It’s the reason that you know things and have goals that none of your individual neurons have.
It arises because individual cells such as neurons have ion channels in their plasma membrane that allow them to generate voltage gradients, which may or may not propagate to neighboring cells. That allows the system to do computations and have goal states and preferences and all these things that individual cells don’t have.
It turns out that this amazing ability—using electricity as a kind of cognitive glue to bind together subunits into a larger system that knows things and solves problems in a different problem space—is extremely ancient. This is not specific to the brain. Evolution discovered something like this around the time of bacterial biofilms.
Every cell in your body has ion channels. Most cells are connected by gap junctions, these electrical synapses that allow them to form electrical networks.
We’ve now developed molecular-level tools to read and write the electrical information in tissue. Just as one might try to read the electrical states of the brain to decode cognition, you can do the same with the body.
For example, in an early frog embryo, using a voltage-sensitive fluorescent dye, we can see what we call the “electric face.” This was discovered by Danny Adams in my lab. Long before the face becomes regionalized into eyes, mouth, and so on, the anterior neural ectoderm carries a bioelectrically encoded map of what the future face is going to look like. If you can read out those voltage gradients, you can literally see where the eye is going to form, where the mouth is going to form, and so forth.
What we’ve found over the years is that these standing bioelectric patterns within the body encode large-scale anatomical features.
For example, in planaria, these flatworms—which are important model systems for aging research—there’s a voltage gradient that allows every fragment to regenerate properly. If you cut a worm into pieces, a middle piece normally makes exactly one head and one tail.
We can go in and reprogram that electrical gradient. We can convert it into a pattern that says “two heads.” The animals remain anatomically normal and molecularly normal, but when you cut them, they regenerate into two-headed animals. These are real animals. That means the set point toward which all the cells are working when they regenerate has been respecified to say “two heads.”
So control of head number, and control of large-scale body plan, is stored electrically. There’s nothing genetically wrong with these animals. We didn’t edit the genome. This is a physiological layer of information.
Another example is using these same signals to ask tissue to grow new organs. In a tadpole, we injected an ion channel that induces a particular bioelectrical state, causing cells to receive a “build an eye here” signal. We’re not talking directly to stem cells or telling them how to build an eye. We have no idea how to build an eye. What we do know is the high-level prompt that, when established, is interpreted by the cells as “build an eye here.” And that’s what they do.
Leo, do you want to talk about this part?
Leo Lopez:
Yes. As Michael introduced it, if bioelectricity is an interface to high-level anatomical control, we wanted to know what happens to these bioelectrical patterns during aging.
So we wrote a review-perspective paper in 2024, where we surveyed the literature on ion channels, gap junctions, and aging.
We found many links, and in that paper we made three main hypotheses.
The main one is that aging is due, or partly due, to the corruption of the bioelectric pattern of the tissue. During aging, the pattern becomes more blurry or degraded.
The second hypothesis is that aging is due to the loss of cellular competency—the ability of cells to interpret the bioelectric pattern—or both, creating a negative feedback loop that leads to aging.
Part of the answer has been realized in the lab: we found senescence-associated membrane depolarization. Meaning that if aging involves an accumulation of senescent cells, then we should also see corruption of the bioelectric pattern associated with that depolarization.
We also found a very specific bioelectric pattern in immortal hydra compared with mortal hydra. So for us now, electricity is definitely involved in aging.
Now we’re going to move more into the loss-of-goal-directedness theory.
We are starting by introducing how we see biological systems: through a cognitive lens. We see biological systems as composed of multi-scale competency architectures. That means cognition exists at multiple levels of biological complexity.
Here, cognition does not mean high-level consciousness. It means cybernetic capabilities: problem-solving, memory, learning, anticipation. We can find those across many levels, from chemical networks to neural networks. Cells can learn and memorize. So with this view, we have a spectrum of cognitive levels.
By modeling biological systems as cognitive systems, we think we gain better predictive and control capabilities. We use the cognitive lens to define the sophistication of the agent and the scale of its goals. This is what we mean by goal-directedness.
Michael Levin:
In the first part of the talk we mentioned these bioelectric patterns and how they define specific structures in the body. I now want to talk about how cells interpret these patterns.
I’m going to make the claim that these patterns represent biophysical encodings of goal states that the cellular collective tries to implement.
It’s very important that we understand the storage, interpretation, and potential degradation of those goal states, because they drive first embryonic development and then maintenance of the body throughout adult life.
To start talking about goals in living systems, we have to understand that goals are not just for complex brains and not just for behavior in three-dimensional space. Biology has a multiscale architecture where every level of organization can do things recognizable to behavioral scientists.
For example, it is now seen that even molecular networks—never mind cells, but pathways within cells—are capable of several kinds of learning, including habituation, sensitization, and even associative conditioning. We are trying to take advantage of that in our lab to train pathways for things like drug conditioning.
Now we come to a critical part for aging. This is a nice version of the Ship of Theseus analogy. Over time, all the planks of the ship are replaced. Is it still the same ship?
People use this analogy for the body. Materials come and go, cells age and die and are replaced, yet the organism persists. Puberty and other changes happen, but overall the large-scale coherence remains.
The interesting thing is that the ship isn’t the physical boat. The ship is actually the pattern in the mind of the replacement machinery—the workers. As long as the workers have the correct plan and all agree on it, the pattern continues. Materials can flow in and out.
In that sense, we are a process, not a structure. That is fine, as long as everybody has the same plan.
A champion of that process is the planarian flatworm. Asexual planaria show no signs of aging at the level of the organism. Individual cells age and die, but they are continuously replaced and regenerated in place, and the worm just goes on.
So we want to understand how this pattern in the body is stored. We’ve mentioned that bioelectricity is really important. We want to understand whether we can rewrite that pattern, so that when it becomes blurry or degraded, we can reinforce it. And we want to understand what happens when those patterns degrade, and how cells use these electrical networks to align toward common purpose.
No individual cell knows what a body is or how many eyes you’re supposed to have, but the collective absolutely does.
Leo briefly mentioned the notion of a cognitive light cone, which is basically the size in space and time of the largest goal states a system can pursue.
A unicellular organism has small local goals. But something amazing happens during the evolution of multicellularity and embryonic development: individual cells come together to pursue enormous goals.
For example, if you amputate a salamander limb, it grows back a very good copy and then stops. That’s the most amazing thing about regeneration: it knows when to stop. There is a homeostatic set point in anatomical space.
That large-scale set point is possible because cells are joined into physiological networks. And that network has a failure mode: cancer.
What you see in glioblastoma cells, for example, is that when cells disconnect from the electrical network, they roll back to their ancient unicellular self. They can no longer remember the larger pattern they were working on. They are not more selfish—they just have smaller selves. The rest of the body becomes outside environment.
That becomes important in Leo’s story about loss of goal-directedness.
Leo Lopez:
We wanted to understand aging through this multi-scale competency architecture. So we developed a model based on neural cellular automata—basically cellular automata on steroids.
We have a grid of cells, and each cell’s behavior is controlled by a neural network, equivalent to a gene regulatory network. The collective cellular behavior gives rise to system-level behavior, in this case morphogenesis.
The key point is that we evolved these neural cellular automata to perform only development and nothing else. We did not select them for good morphostasis or anti-aging, just for reaching the target morphology—a smiley face.
What we observed is that all the organisms reached the adult morphology, and then we began to see degradation of the anatomy. Sometimes they even lost organs, like the eyes or mouth—basically death.
What is very important is that we have aging in the simulation without any external perturbation or accumulation of damage. Aging emerges as a disorder of collective behavior.
We interpreted this as aging being a loss of goal-directedness in anatomical space. The cells no longer have high-level anatomical goals after development.
In the simulation we also found something interesting: we could reactivate regenerative capabilities. By injecting embryonic states into cells that had lost an organ, we reactivated developmental programs that were basically regenerative programs. So in effect, there was rejuvenation of the anatomy by targeted simulation.
This relates to partial cellular reprogramming, where embryonic factors can induce regeneration or rejuvenation, for example in the eye.
This loss-of-goal-directedness theory also makes predictions:
- Never-ending anatomical goals may lead to minimal aging and lower cancer rates.
- Organisms with continuous growth or whole-body regeneration—such as lobsters, some trees, planaria, and Greenland sharks—may show negligible aging or extreme longevity partly for this reason.
- Continuous anatomical goal-directedness may also help prevent cancer by maintaining the morphogenetic field and cellular alignment, preventing cells from dropping down to smaller-scale goals.
Michael Levin:
To summarize what Leo just said: in the simulation there is no underlying damage. We are not modeling entropy accumulation. We do not have any evolutionary selection for death. And yet, the goal-directed system—a collective of cells minimizing distance from a specific shape—starts to disband after the goal is achieved.
This raises a very deep philosophical question: what does a goal-directed system do after it has met its goal?
It suggests another possible cause of aging, one that has nothing to do with physics or selection, but is fundamentally cognitive.
What about aging in cells?
Leo Lopez:
We wanted to test whether we see an “atavistic” genetic expression pattern during aging, similar to cancer cells.
So we did a phylostratigraphic analysis of many different aging tissues and cell transcriptomic datasets. This means we assign each gene an evolutionary age and ask whether the genes differentially expressed during aging are overrepresented among older or younger genes.
What we found, in two large multi-tissue aging signatures, was an important dissociation: overrepresentation of more ancient genes, and underrepresentation of younger genes, compared with the age distribution of all human genes.
We found this in skin, senescent mesenchymal cells, senescent immune/ovarian contexts, and human prostate cells. We did not find it in brain and stem cells, which may be compatible with differential aging data.
So the way we see aging is that during development all the cells are aligned toward a big goal: reaching the appropriate anatomy. Once development is complete, they start to follow their own smaller cellular goals, leading to misalignment and gradual anatomical decline.
Michael Levin:
To summarize, there are three big ideas here.
First, a major aspect of aging may be the problem of what a cognitive system does after it has met its goal. Cellular collectives are very good at achieving anatomical goals, but once the body is constructed and maturation is completed, the collective begins to disband.
Second, part of that process is the blurring of bioelectric patterns—the memories that keep the system aligned toward a species-specific shape.
Third, we may literally be seeing cells dissociate from one another because they begin to express transcriptional signals that are evolutionarily older. The cells of an aging body are no longer on the same page about what kinds of genes they should be expressing.
Finally, Leo and I have been thinking about aging and longevity beyond the level of the single individual. Longevity isn’t just about maintaining the current standard human body for as long as possible. With bioengineering, morphogenesis control, and other technologies coming online, it is quite possible that humans 50 years from now will not look exactly like current Homo sapiens.
The question of longevity is not just “How long can you maintain the exact body you have now?” It is also: What is it that you want to persist? What do you want to be?
Maybe we can radically prolong a normal human body by giving cells the same goals over and over again. Or maybe they require novelty in the goal state, like a caterpillar becoming something else. That remains to be seen.
At this point, we want to thank the postdocs, students, collaborators, and funders who supported this work.
Host:
That was great. Thanks, guys. I have so many questions. Even though that was a 30-minute presentation, I feel like you could go for hours on many of these segments.
My first question relates to the anatomical compiler, which I’ve heard you talk about many times. It seems like we need to learn to communicate with ourselves to give our tissues that goal-directedness.
My own work focuses heavily on diet and biomarkers, and in a rudimentary way I’m trying to learn what my kidney or liver “wants” by tracking correlations between diet, supplements, and biomarkers. That’s basically precision health or precision nutrition.
So how do you foresee the anatomical compiler going beyond self-tracking and biomarkers into actual communication with organs? You can’t do patch-clamp measurements across a whole human. So how does this translate into people?
Michael Levin:
A few things. First, I should disclose that there is a company called Morphoceuticals, spun out of our group, which is developing some of these technologies.
Our goal absolutely is to get good bioelectric signaling in large-scale organisms such as people. There are technologies we hope will be developed soon to allow that.
The point of the anatomical compiler is that it is a communications device. One of the things we’re doing is using AI, VR, and other tools to develop ways to literally communicate with your organs.
So when you talk about finding out what the liver and other organs want, I think that is actually doable. These things are not passive machines. They are problem-solving agents that continuously navigate complex physiological state spaces. They do have goal states. They do “feel” stress when deviated from them. Those are things we should be able to communicate with—if we can crack the code and talk to them on their own terms.
Bioelectric interfaces are the most obvious, but there are probably others: biomechanical, optical, and so on.
This has to start in model systems, but I firmly believe the fundamentals are evolutionarily conserved. When we figure out how to talk to planarian cells, frog cells, and mouse cells, we will have tools for figuring out what kinds of goal states human cells need in order to do what planarian cells clearly show is possible.
Leo Lopez:
At the end, the question is where the goals are encoded and how we can rewrite them. We have shown in the lab that this can work with bioelectricity in planaria, but for humans we still need more data.
Host:
Would this involve imaging the whole human to detect membrane potentials across organs? What technology would you use?
Michael Levin:
Right now, the voltage-sensing technology is optical. There are dyes being developed for this. We hope to develop similar reagents for CT, MRI, or ultrasound, so you can go into deep tissues and get voltage maps.
But something important here is that you probably do not need a single-cell-resolution map of what every cell is doing, because that’s not how the code works. The code is not a single-cell code. It is a coarse-grained code in which large-scale patterns matter.
That is what we’ve been trying to understand for about 25 years: what is it about bioelectric states that other cells are actually interpreting?
We also have another spin-off called Astonishing Labs, which is looking at various aspects of aging in general.
Host:
Would one time point be enough, or would you need multiple measurements over time and across physiological states?
Michael Levin:
This is what we are figuring out now in model systems. There are signals at multiple timescales. Some bioelectric signals will reflect metabolism or transient physiology, which are not what we’re looking for. Others will be slow-changing gradients that say, for example, “this is what your kidney should be shaped like.”
We had an interesting case where we successfully sharpened degraded voltage gradients in the context of birth defects. We could get normal brain development despite nasty genetic mutations. Later we tried the same for gut and heart defects even though we didn’t have good voltage maps of those tissues, and it still worked.
So there may be conserved general principles we can exploit even without having all the detailed information at once. But we won’t know until we do more complete mapping in model systems and eventually humans.
Leo Lopez:
Yes. This is an empirical research program. We do not know exactly yet. More time points would probably be better.
Host:
The challenge is that there seem to be very few labs using this approach for aging and rejuvenation. What kind of timeline do you foresee?
Michael Levin:
I can’t give exact timelines. It depends on the science, funding, and regulation. But I don’t think it will take 100 years.
And to be clear, there are other excellent labs doing bioelectricity research—people like Mansi Srivastava, Emily Bates, and others—and others studying regeneration and aging. I think all of this is going to converge.
Actually, I think one of the biggest blockers is conceptual. People trained in molecular and cell biology often have a kind of phobia about using the tools of cognitive science to understand what cells and tissues are doing.
Outside the diverse intelligence community, people think it is weird when we talk about cell learning, goal states, motivation, and wiping priors. But the ice is melting. We can now publish and discuss these ideas in ways that were basically impossible a few years ago.
It’s going to happen, and I don’t think it’s going to take a hundred years.
Host:
One rabbit hole is whether fixing the bioelectric patterns in the brain might be the biggest lever. If you fix them there, does everything else follow?
Michael Levin:
You’re asking two related questions.
One is to what extent signals from the brain are actually important for managing the rest of the body. There is some evidence for that—for example, the nervous system has roles in limb regeneration, and some aspects of development don’t go quite right without a brain.
But by and large, you do not need the brain to do most of morphogenesis. A huge amount of morphogenesis happens before the brain even forms. So it is not clear that you need the brain for the kind of tissue upkeep we’re talking about.
The second question is maintenance of the brain itself. A major open question is whether cognitive changes with aging are primarily a hardware problem or a software problem. If we kept rejuvenating the cells, would we retain the plasticity of a 20-year-old? Or after some number of years does a human cognitive architecture just get stuck in certain ways?
We do not know.
Leo Lopez:
Yes, this is a real question. It is not clear whether the brain changes of aging are hardware, software, or adaptation.
But the broader point is that we are talking about collective intelligence of the body, and if we can reactivate developmental or regenerative programs, that may be the equivalent of rejuvenation.
Host:
There is also the planaria argument. You can cut them into many parts and they regenerate, including a brain, and some memories may be encoded somatically. Does that imply you might someday be able to retrain the brain in Alzheimer’s disease from the body?
Michael Levin:
We do not yet know whether mammals or humans can do the amazing trick planaria do—regenerate memories along with brains.
I also think the standard story of how memories are stored in the brain may not be complete. One of the most interesting clinical datasets here is the rare human cases of extremely reduced brain volume with normal IQs. Karina Kaufman and I reviewed these recently. Some people have amazingly little brain tissue and yet overtly normal IQs and personalities.
So the mapping between neural real estate and cognitive performance is not obvious.
And there are other mysteries. For example, general anesthesia disconnects electrical synapses in the brain, yet most people wake up as themselves. That already tells us there are forms of stability we do not understand very well.
Host:
Then there is planaria themselves. They seem to maintain this regenerative, rejuvenative cycle indefinitely. How can we use that in people?
Leo Lopez:
Planaria probably have never-ending anatomical goals—constant regeneration. That may explain why they do not age, at least according to our simulations and this framework. The idea would be to do something similar in mammals one day, but how exactly remains an open question.
Michael Levin:
Planaria tear themselves in half every couple of weeks and create a new regenerative challenge. It is possible that if we increased regenerative capacity in humans, that could refresh these goals.
One interesting thing is that planaria have very noisy genomes. Because they reproduce by tearing themselves in half, they keep somatic mutations instead of resetting them each generation the way we do. Their cells can even be mixoploid. And yet they have extraordinary anatomical fidelity, regenerative capacity, cancer resistance, and practical immortality.
That suggests that when evolution cannot rely on the hardware being clean, it can improve the algorithm—the communication and coordination between cells—so the system becomes highly tolerant of problems in the hardware.
There is a whole spectrum here. Species differ in how good they are at improvising solutions from strange starting points. Planaria are far to the high-plasticity end.
Host:
You’ve also mentioned the barium experiments, where planaria initially fail and then adapt. Does that mean they are better at computation than human cells?
Michael Levin:
I would not make that argument. I think all cells compute. There may be differences, but very little of this has been studied.
For example, Doug Blackiston in my lab made tadpoles with eyes on their tails. Out of the box, those animals could still use those eyes for visual behavior. The eyes did not even necessarily connect to the brain in the usual way. Sometimes they connected to the spinal cord, sometimes the gut, sometimes nowhere obvious. Yet the animals could still perform visually guided tasks.
Why does that work? Because evolution does not make solutions to one fixed environment. It makes problem-solving systems.
Host:
Is the future of human regeneration then using bioelectricity to guide development and regrowth, rather than bottom-up gene editing approaches?
Michael Levin:
There are two questions there. First, does a top-down approach exist at all? Second, if it does, is the top level bioelectric or something else?
Here is what we know. We and others have already demonstrated many cases where a simple high-level intervention triggers a complex morphogenetic program.
For example, in frog leg regeneration, 24 hours of stimulation with a simple sodium ionophore led to a year and a half of leg growth. We didn’t tell the tissue how to build a leg. The cells knew what to do.
Likewise for inducing eyes: we do not know how to build lens, retina, optic nerve, and all the rest. We only know how to issue the prompt “build an eye here.”
So top-down programming clearly works.
Is bioelectricity the top level? I think so, because we have shown that with simple bioelectric patterns, downstream biochemistry, gene expression, cell movement, epigenetics, and other processes line up. There may be something upstream of bioelectricity, but we haven’t seen it clearly yet.
Leo Lopez:
The future of medicine may be in top-down control. Planaria are genetically messy but still regenerate. Mammalian cells may be more genetically stable, but they do not regenerate. So perhaps what matters is the algorithm or software, and this brings us back to top-down programming.
Michael Levin:
I’ll end with a brief story about what might even be upstream of bioelectricity.
A physician named Albert Mason developed a field sometimes called hypnodermatology. In the 1950s in the UK, he used hypnosis in place of anesthesia for childbirth, and later used suggestion to treat severe skin conditions. He reportedly got dramatic results.
But then he noticed that although patients’ skin problems improved, other life problems emerged—gambling, marriage problems, and other behavioral issues. He concluded that he had intervened at a level above drugs, but was still just moving the problem around. So he moved into psychiatry, trying to address the deeper cognitive issues that might be expressing through somatic symptoms.
That raises the question: even if bioelectricity is a top-down level, is it actually the highest level? Possibly not.
That is why in my group this work is tightly integrated with fundamental questions of cognition, memories, goal states, and so on. In the end, I do not think the important thing is the physics of bioelectricity per se. I think what matters is that bioelectricity may be implementing a deeper kind of cognition in cellular collectives.
Host:
I think you guys are hard out, so I don’t want to keep you, but I have so many other questions—anthrobots, xenobots, metabolomics of rejuvenation, axolotls, deer antlers, human stem cells. Hopefully we can do this again.
Michael Levin:
Absolutely. Let’s talk again.
Host:
All right. Thanks very much.
Summary
This conversation presents a highly unconventional but intellectually coherent framework for aging and rejuvenation.
The core argument is:
-
Aging may not just be damage accumulation or bad genetic programming.
It may also be a failure of higher-order coordination in tissues. -
Bioelectric patterns act as a kind of tissue-level control layer.
These patterns are proposed to store or transmit “goal states” that tell cell collectives what large-scale anatomy they should maintain. -
Development and regeneration show that cells can follow high-level instructions.
Levin’s examples include planaria forming two heads after electrical reprogramming and tadpoles growing ectopic eyes after bioelectric manipulation. -
Aging may result from loss of goal-directedness after development is complete.
Once the organism reaches adult form, the multicellular collective may no longer maintain a strong shared anatomical objective, so cells drift back toward smaller, local goals. -
Cancer is framed as a failure of cellular integration.
Cells disconnected from the larger bioelectric and morphogenetic network revert to a more primitive unicellular style of behavior. -
Regeneration and rejuvenation might come from restoring or rewriting the tissue’s high-level control state.
In this view, interventions like gene editing and epigenetic reprogramming are downstream. Bioelectric control might sit upstream of many familiar molecular mechanisms. -
The speakers also gesture beyond bioelectricity.
Levin suggests bioelectricity may itself be a mechanism for implementing something deeper: cognition, memory, or goal representation in living systems.
Novelty
The most novel aspects are these:
1. Aging as a cybernetic failure
Rather than treating aging mainly as molecular damage, the speakers propose that aging is partly a control-systems problem: a failure to preserve high-level anatomical goals.
2. Bioelectricity as an instructive layer for morphology
They do not present bioelectricity as merely one signaling modality among others, but as a higher-order patterning system that can encode large anatomical outcomes.
3. “Loss of goal-directedness” as a theory of aging
This is the conceptual centerpiece. The idea that development succeeds because tissues have a strong shared objective, but adult aging begins when that objective is exhausted or degraded, is highly original.
4. Cancer as shrinking of the cellular “self”
Their reframing of cancer is also unusual: cancer cells are not just selfish or mutated, but become disconnected from large-scale goals, reverting to smaller-scale ones.
5. Computational modeling of aging without damage accumulation
The neural-cellular-automata result is conceptually interesting because it suggests aging-like decline can emerge in principle even without explicit damage terms, simply from post-developmental loss of coherent system-level control.
Critique
What is strong
1. The framework is genuinely integrative
One of the best things about this talk is that it tries to unify:
- development,
- regeneration,
- cancer,
- aging,
- and tissue maintenance
under a single conceptual umbrella.
That is ambitious, but not frivolous. Many mainstream aging models explain decline, but do a poorer job of explaining why complex anatomical order is maintained at all, or why regeneration is possible in some species but not others.
2. The regenerative examples are important
The planaria and tadpole examples support a real and important point: cells can respond to relatively high-level cues and then execute astonishingly complex structures without humans specifying every molecular step.
That general lesson is credible and valuable.
3. The emphasis on pattern-level biology is probably underappreciated
Biology is often over-reduced to genes, proteins, and cell-autonomous pathways. The talk usefully pushes back by emphasizing:
- tissue-level patterning,
- cell-cell communication,
- emergent coordination,
- and non-genetic information storage.
Even if the strongest version of their theory proves wrong, this emphasis is likely fruitful.
4. Their language helps generate testable questions
A lot of big-theory talks fail because they only sound profound. This one at least suggests some concrete research directions:
- map bioelectric patterns during aging,
- test whether restoring them improves tissue function,
- ask whether senescent cells disrupt tissue-level electrical states,
- compare regenerative versus non-regenerative organisms,
- test whether tissue maintenance can be strengthened without changing DNA sequence.
That is a real scientific program.
What is weak or speculative
1. The leap from regeneration biology to human aging therapy is enormous
The talk is strongest in planaria, frog embryos, and regeneration models. It is much weaker when implying that the same framework is close to explaining or treating human systemic aging.
There is a very large gap between:
- showing that bioelectric signals help pattern embryos or regeneration,
and - showing that aging in adult humans is meaningfully driven by loss of these same signals in a therapeutically tractable way.
That bridge has not yet been built.
2. “Goal-directedness” risks being metaphor-heavy
The language of goals, cognition, and memory is stimulating, but it also risks becoming too elastic.
A scientific danger here is that terms like:
- goal,
- intelligence,
- memory,
- preference,
- motivation
can shift from precise operational meaning into broad metaphor. If not tightly defined, the framework can become difficult to falsify.
The theory is most persuasive when translated into measurable claims such as:
- stable distributed control states,
- error-correction toward target morphology,
- large-scale voltage gradients,
- network integration across tissues.
It is least persuasive when it sounds quasi-philosophical.
3. The computational model may demonstrate plausibility, not reality
The neural-cellular-automata model is interesting, but it shows that aging-like decline can emerge without damage, not that this is what does drive aging in real organisms.
A toy or abstract computational model is useful for generating hypotheses, but it does not by itself weigh strongly against damage-based explanations of aging in mammals.
4. Some species examples are suggestive but not decisive
The appeal to planaria, lobsters, trees, or Greenland sharks is intriguing, but comparative biology is messy. These organisms differ from humans in many dimensions:
- body plan,
- reproduction,
- stem cell dynamics,
- cancer suppression,
- metabolism,
- ecology,
- and developmental architecture.
So the existence of negligible senescence elsewhere does not, by itself, strongly validate the goal-directedness theory.
5. The relationship to known hallmarks of aging remains underdeveloped
A stronger version of the talk would have more explicitly connected the theory to mainstream hallmarks such as:
- genomic instability,
- mitochondrial dysfunction,
- epigenetic alteration,
- cellular senescence,
- stem-cell exhaustion,
- altered intercellular communication,
- proteostasis failure.
Right now, the framework tends to imply these are downstream, but it does not yet clearly show how bioelectric degradation mechanistically produces them, or vice versa.
6. The final “maybe something upstream of bioelectricity” move weakens the hard claim
Levin is admirably honest in acknowledging that bioelectricity might not be the ultimate top layer. But this also makes the framework more difficult to pin down.
If bioelectricity is the master regulator, that is a bold and testable thesis.
If bioelectricity is just one visible implementation of deeper cognitive organization, the theory becomes broader but also less sharply testable.
Bottom line
This is a highly original, intellectually rich, and provocative talk. Its biggest strength is that it forces people to think beyond a purely reductionist view of aging and to take seriously the possibility that tissues maintain anatomy through distributed, information-rich control systems.
Its biggest weakness is that it often moves too quickly from:
- fascinating regeneration biology,
to - very large claims about aging, cancer, cognition, and human rejuvenation.
So the talk is best viewed as:
- not a proven theory of aging,
- not a near-term clinical roadmap,
- but a serious and creative research framework that may eventually illuminate parts of aging biology that standard molecular models miss.
My overall judgment:
Scientific value: high
Immediate evidential strength for human aging: moderate to low
Conceptual originality: very high
Risk of overextension/speculation: high
If useful, I can also turn this into a three-column table with claim / evidence cited in the talk / critique.