I went through a bottle of Boswellia (Double Wood) for my knees, but nothing.
The problem is that the Mayo Protocol on Fisetin failed. They tested it and got nil/negative results. That’s why they dropped it like a hot potato. Fisetin wasn’t worth them spending any more time, energy or money on.
“Our findings argue against what many people are already doing — using commercial products like quercetin or related compounds like fisetin that may show some senolytic properties,” says senior author Sundeep Khosla, M.D., an endocrinologist at Mayo Clinic in Rochester, Minnesota. “They’re using them as anti-aging agents without knowing if they have high enough senescent cell numbers to benefit, or what dose or dosing regimen is needed to be effective yet safe.”
It is better to prevent senescent cells from forming with a senomorphic such as Rapamycin or Taurine instead of allowing them to form and removing them later.
IMO, frankly we have very little idea of what “senescence” actually is, or how it is contributing towards ageing as a whole. There isn’t only one single senescent phenotype. Different cell types show different responses to stress, and secrete different SASP. Senescence is useful in many situations, including preventing tumour formation. So it’s a stretch to then imagine that preventing or killing them is beneficial. I’d also point out that I think reproducibility in the field is pretty low, and a lot of the models (like using H2O2 or staurosporine etc to induce senescence) are not very useful.
This includes the quote you posted: https://newsnetwork.mayoclinic.org/discussion/drugs-that-kill-zombie-cells-may-benefit-some-older-women-but-not-all-mayo-clinic-study-finds/
Dr. Khosla was discussing the D+Q trial, which found the combination beneficial to bone metabolism. Although vague, he seems to be comparing the range of supplements, including fisetin and quercetin, that can be bought over the counter, with D+Q, finding D+Q to be better. That D+Q is stronger medicine than fisetin isn’t a matter of controversy.
As relaxedmeatball says, cellular senescence isn’t well understood. It is a real thing, though, and antecdotal evidence is enough for some of us to give this or that intervention a try. As is the case here.
After trying out the Mayo fisetin protocol a couple of years ago and discovering that the brand I was using, Vitablossom, didn’t contain fisetin, the recent D+Q posting encouraged me to try it again, this time with a good brand, Toniq. I just finished the first round.
Lots of articles on senolytics out there, so you’re probably referring to one that specifically finds fisetin useless. Could you provide a link to the Mayo trial you’re referencing?
I was interested in Fisetin and took it based on the Mayo Clinic trials back in 2022. I was constantly waiting for the results to be published. I waited and waited. They said it would be any month now. 3 years later and still nothing. If there were any positive results, they would have published something by now. When all goes silent, it’s a sign of failure.
The only thing Fisetin did for me was give me bad diarrhea. Then I sarted Rapamycin and Taurine and never looked back.
Also, Aubrey DeGrey’s mouse study tried a pharmaceutical senolytic which actually decreased mouse life expectancy. Senolytics may not extend lifespan.
I see your point and have had the same thoughts. Dunno.
I’m going with the positive anecdotal evidence for now and proceeding. The sudden, crippling arthritis was eye-opening. There’s no doubt that restarting rapamycin took care of all but some remnants. Maybe I’m being greedy, but I want those remnants gone too.
“OA was artificially induced (ACL rupture), whereas in primates it developed naturally with aging.” This is a bigger deal, a major confound in my opinion, to equate artificially induced rupture with OA. Making it a struggle to have confidence in the conclusions. With an acute injury there is a major inflamatory response, which helps splint the joint, and in practice we would likely stop the rapamycin till the acute injury is improved, like I did for 1 yr after my knee replacement.
Didnt realize it was artificially induced. I am sure this model has been validated or is it not?
But then in the context of healing/ repair of somwhat less vascular tissue with Rapa on board does make me wonder about the study design. I am sure the investigations thought it through.
Does glucosamine confer any protection against OA in this setting?
According to the NYT, new treatments for osteo, delivered by injection, can regrow bone and cartilage. Growing whole knees may be next.
Federal Agency Unveils Three Potential Osteoarthritis Treatments
With funding from ARPA-H, three teams of researchers have regrown bone and cartilage, even entire knees, in animal studies. Human trials are not far off.
Listen · 6:47 min

A colored X-ray of a 66-year-old patient with osteoarthritis.Credit…Dr. P. Marazzi/Science Source
By Gina Kolata
April 6, 2026Updated 5:40 p.m. ET
Three years ago, a little-known new federal agency announced its first big project: It would invest tens of millions of dollars over five years to find a cure for osteoarthritis, the painful wearing away of joints that affects 32 million Americans.
Now the agency, ARPA-H (the Advanced Research Projects Agency for Health), says it has several promising solutions. Its research teams are contractually obligated to start testing in patients within 18 months.
The new research focuses on knee osteoarthritis, but the investigators believe the solutions could eventually be applied to osteoarthritis in any joint.
Two research teams, at Duke University and at the University of Colorado, Boulder, have developed injections or infusions that regrow bone and cartilage, ARPA-H announced on Monday.
And a third team, at Columbia University, may have discovered a way to regrow an entire knee.
The methods so far have been tested only in animals. But experts said they were encouraged by the findings.
“It’s hugely promising,” said Dr. Scott Rodeo, vice chair of orthopedic research at the Hospital for Special Surgery in New York, who was not associated with the studies.
“Right now, everything we have just modifies symptoms,” he added. Curing arthritis by regrowing cartilage and bone “would be a paradigm shift.”
Finding a new approach is the point, said Dr. Ross Uhrich, program manager for the osteoarthritis project at ARPA-H. “If your goal is just to publish a journal article, this is not the agency for you,” he said.
ARPA-H is part of the Department of Health and Human Services. It is modeled on a similar program at the Defense Department, DARPA (the Defense Advanced Research Projects Agency), which helped develop the internet, GPS and autonomous drones, among many other technologies.
At ARPA-H, the scientists who applied for the arthritis funding, chosen from a range of companies, labs and teams, agree to solve the problem in preclinical and clinical studies. If their treatments are successful in humans, they must commercialize them.
The investigators also must agree that their arthritis products will be tested in the groups most likely to need it, meaning that more than half of the clinical-trial participants must be women and the trials must include Native Americans and Alaska Natives.
If a treatment is approved for marketing, it must cost no more than 25 percent of the price of the current standard of care.
Columbia University’s team leaders, the biomedical engineers Clark Hung and Nadeen Chahine, say they’ve found a way to regrow a knee using an artificial joint made of a 3-D printed “scaffolding” filled with bone and cartilage cells.
Those cells grow into healthy bone and cartilage while the scaffold slowly dissolves over about a year. The treatment is meant for so-called bone-on-bone patients who have lost all or nearly all the cartilage in their knees.
The cells that fill the scaffold can be derived from the patient’s abdominal fat or banked donor cells. If the cells are taken from abdominal fat, the investigators isolate stem cells and then use biological modifiers to direct them to morph into cartilage and bone cells — a process that takes a month.
Donated banked cells, on the other hand, don’t have to change into bone and cartilage cells, Dr. Chahine said. In the scientists’ experiments, these cells did not provoke an immune reaction despite their origins.
To see if the scaffold would act like a real knee, weight-bearing and flexible, surgeons implanted their experimental knees in cadavers and used robotics to test the joints’ suitability for walking.
The next step is to test the system in large animals, Dr. Chahine said. Their five-year ARPA-H contract supplied the team with a bit more than $42 million. “For academic research, this is like pie-in-the-sky money,” she added.
A federal grant is typically a little more than half-a-million dollars. But with a grant, no one is expected to take their work from theory all the way to marketing approval.
At Duke, Dr. Benjamin Alman, an orthopedic surgeon, said the team’s idea was to ask if they could get a patient’s cartilage cells to regrow.
At the very end stage of osteoarthritis, there are none left. But a vast majority of osteoarthritis patients have some cartilage cells in their knees. Could they make the cells start dividing and repopulating the knee with healthy cartilage?
In osteoarthritis, bone gets thicker, changing the mechanics of the joint. Could they remodel bone to make the knee normal?
The researchers tried a variety of medicines alone and in combination, and they ended up with three treatments.
One, an injection, makes cartilage cells grow. Another shot remodels bone. The third, an infusion that can treat several arthritic joints at once, also makes cartilage grow.
“The idea would be, if a patient’s problem is mostly in cartilage, we would target cartilage,” Dr. Alman said. “If it is mostly bone, we would target bone.”
The treatments have worked in rats and mice. “I tend to be very skeptical, but this surprised me,” Dr. Alman said.
Stephanie Bryant, a chemical and biological engineer who heads the team at the University of Colorado, Boulder, said her goal was “to return the tissue to a healthy state,” with at most one injection.
The group found a drug, a repurposed medicine already on the market, that worked in animals and developed a formulation that releases it in bursts over months. The drug, she said, “resides long-term in the joint.”
Their tests included rabbits that got the equivalent of an A.C.L. tear in humans. Like humans, they quickly developed osteoarthritis. Within two months after the injection, their knees had regenerated.
The other animal model was a guinea pig that develops the sort of degenerative arthritis that often happens as people age. Once again, the treatment seemed completely effective for the knees.
But what about people with advanced bone-on-bone arthritis? The Colorado group has developed a mixture of engineered proteins that are injected into the joint and fill in the space where cartilage should be.
Those proteins recruit so-called progenitor cells from the underlying bone and induce them to regrow. Among the group’s rabbits, it took just three months before all the injected material was gone. What was left was healthy cartilage.
The team, Dr. Bryant said, was overjoyed. “You do all of this work, have all these hypotheses, but you never know if it will work,” she said.
“Very few of us get the opportunity to really push forward things we are working on,” she added. “This is an opportunity to really help patients.”
Gina Kolata reports on diseases and treatments, how treatments are discovered and tested, and how they affect people.
Why should it be? I have osteoarthritis and rapamycin has not helped at all.
I asked Gemini 3 Pro what this drug was that they were likely referring to. Here is the response:
Investigative Report: ARPA-H NITRO Program and the Repurposing of Teriparatide
Target Drug Identification
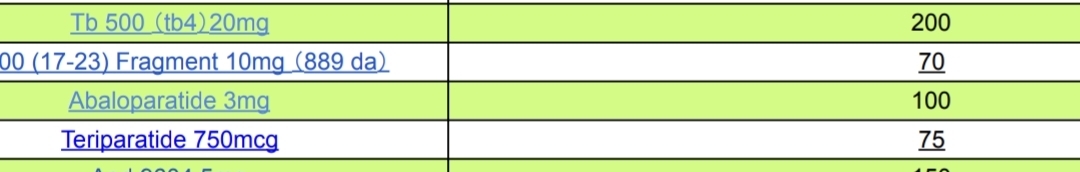
The repurposed medicine referenced in the source text is Teriparatide (commercially available under the brand name Forteo), a recombinant human parathyroid hormone (PTH 1-34) analog. An alternative candidate in the same class currently under investigation by the research group is Abaloparatide (Tymlos).
The Investigators and the ARPA-H Framework
The research is funded by the Advanced Research Projects Agency for Health (ARPA-H) under its Novel Innovations for Tissue Regeneration in Osteoarthritis (NITRO) program. The primary investigators matching the criteria are:
- Dr. Stephanie Bryant: Professor of Chemical and Biological Engineering at the University of Colorado Boulder. She specializes in biomaterials, tissue engineering, and hydrogel scaffolding.
- Dr. Michael Zuscik: Professor and Vice Chair of Research in the Department of Orthopedics at the CU Anschutz Medical Campus. Zuscik has spent over 15 years investigating the biological application of PTH analogs for osteoarthritis.
- Commercial Entity: To satisfy the ARPA-H commercialization mandate, the lead researchers recently launched Renovare Therapeutics, Inc. to advance these candidates through Investigational New Drug (IND)-enabling studies.
Mechanism of Action and Longevity Pathways
In standard clinical practice, teriparatide is administered via daily systemic subcutaneous injections to treat severe osteoporosis by stimulating osteoblast-mediated bone formation. However, Zuscik’s laboratory established that the activation of the parathyroid hormone 1 receptor (PTH1R) is also highly chondroregenerative.
In the context of post-traumatic and age-related osteoarthritis, PTH1R is upregulated in damaged joint tissue. Agonists like teriparatide push chondrocytes (cartilage cells) out of a degenerative, catabolic state and into an anabolic state. This pathway stimulates the synthesis of the extracellular matrix—primarily Type II collagen and proteoglycans—effectively reversing structural degradation and promoting joint healthspan.
The Formulation Breakthrough
The primary limitation of using teriparatide for joint regeneration has historically been delivery mechanics. Daily systemic injections are impractical for localized joint wear and pose off-target risks, leading to poor patient compliance. Dr. Bryant’s engineering team solved this by developing a proprietary polymeric particle delivery system. This biomaterial acts as a localized carrier that is injected directly into the joint space, releasing the PTH analog in sustained, intermittent bursts over several months. This localized pharmacokinetics profile allows the drug to reside long-term in the joint, successfully repairing ACL-tear models in rabbits and age-related degenerative arthritis in guinea pigs.
Scholarly Debates and Knowledge Gaps
While the preclinical data presents a paradigm shift in geroscience and orthopedics, several critical knowledge gaps remain before this can be validated in human clinical applications:
- Translational Efficacy: Cartilage regeneration in murine, rabbit, and guinea pig models is not a guaranteed predictor of human efficacy. Human joints endure significantly different biomechanical loads, have distinct cartilage thickness, and exhibit different basal metabolic rates.
- Release Kinetics and Pharmacodynamics: The exact degradation rate of the proprietary hydrogel/nanoparticle carrier in a human synovial environment is currently unknown. If the carrier degrades too rapidly, a localized overdose of PTH could theoretically trigger ectopic bone formation (osteophytes) rather than uniform cartilage regeneration.
- Long-Term localized Safety: Sustained PTH1R activation carries theoretical risks of localized cellular hypertrophy. While the FDA has removed the black-box warning for osteosarcoma on systemic Forteo, long-term intra-articular exposure requires rigorous monitoring.
To provide a conclusive assessment of this therapy, the publication of Renovare Therapeutics’ impending Phase 1 human safety data, alongside the exact molecular specifications and release kinetics of their delivery carrier, is required.
Has anybody tried PRP injections to treat OA? Or HA injections?
Aprox 12 years ago I had PRP in my knees, lower back, and neck. It helped my lower back.
I had a couple of rounds of HA in my knees aprox 20 years ago. They did help but the pain from the injection, for me, was not worth the short term benefit. I don’t need it anymore, but if I did, I would not hesitate getting them again for a short term need (think hiking trip etc)
20 years ago and don’t need it anymore? So did you manage to cure OA? It’s great! I was under impression that it’s not curable.
I had terrible OA in my elbows and shoulders maybe 15 yrs ago. Doc used cortosol injections and they worked and he assured me it was OA and there’s no cure. I used PRP Once on each joint and all are healed now. It’s been a couple years and I can throw again and use my joints again without restriction. My wife got it in her hands and she is back to milking goats by hand occasionally. She had terrible pain. I think it’s important to find someone with a good reputation and record of success because there are lots of people out there with the equipment to do it and not the knowlege. Don’t go for the bargain.
For clarity, the PRP didn’t work on my knees and the injections only gave me short term relief
I actually went to see an orthopedic surgeon, the one who gave me injections, to discuss knee replacements when I was aprox 40yo. I have chondromalacia patella in both knees and have since my 20s (I remember it hurting going downstairs because I lived on the 2nd floor).
He said you are too young because replacements don’t last forever and the results are not as good the second time. (Perhaps it’s different these days)
Aprox 8 ish years ago I met my PT and he’s done a ton of things to help… he changed the way I move and worked on strengthening everything around my knees.
It took years but I miraculously now only very very rarely have pain outside of my workouts with him.
At times my knees hurt when he’s making me do something, but he’ll quickly just adjust my position and it goes poof. He forces me to do squats etc that I could not even do two years ago.
So, it’s still there but I have just worked to stop triggering it and making it hurt. There is hope!
I also have it in my hand - both thumbs. Per Xray it’s mild, but it hurts a lot if I use hands. Have an appointment to evaluate it. I won’t do corticosteroids, but want to try PRP and HA.
Try walk backwards 10 min a day!
I think OA is one of those diseases that occurs as a result of stem cells not turning properly into somatic cells and going senescent. There is some evidence that links OA to senescence.
Bottom line (from the detailed question response linked to above)
The research supports a credible model in which senescent joint cells contribute to OA by secreting inflammatory and matrix-degrading SASP factors, spreading senescence to neighbouring cells, impairing repair, and promoting cartilage loss plus subchondral bone and synovial pathology. The strongest evidence is from human tissue association and animal intervention studies. The weakest point is clinical translation: senolytics have not yet produced a convincing approved OA therapy, with UBX0101’s failed Phase 2 trial being the major cautionary result.
my words again
I think the reason cells go senescent is a shortage of acetylation at the key differentiation stage and therefore if you wish to fix this that is a key target.
Stimuli such as exercise, nutrition including boron is important, but without the cells making the cartilage (chondrocytes) there will be a problem.
Incidentally I don’t think killing the senescent cells is a good idea. Instead they need to be made functional so that they can do their job of producing cartilage. Hence I am not surprised that interventions trying to kill the cells (senolytics) have not been positive.