USC (University of Southern California):
Why do some people age faster than others? USC President Kim and global aging expert Eileen Crimmins – the Professor of Gerontology at USCLeonardDavis. Explore the science behind healthspan, longevity, and the limits of biohacking.
I. Executive Summary
This transcript details a high-level population health and gerontological analysis by Dr. Eileen Crimmins, focusing on the epidemiological realities of human aging, demographic disparities, and the biological validation of social determinants of health. The core thesis rejects the fragmented approach to age-related disease (e.g., treating cardiovascular disease and cancer as distinct entities) in favor of targeting a universal set of molecular and cellular changes—the hallmarks of aging—that underlie all late-life morbidity.
Crimmins presents a stark critique of the current state of consumer longevity interventions (“biohacking”). She identifies a massive translational gap between population-level epigenetic research and individualized, direct-to-consumer anti-aging protocols. Notably, she warns that single-individual longitudinal tracking using current epigenetic clocks yields unacceptable noise levels—fluctuating by 8 to 10 years within a single subject over mere months—rendering short-term clinical decision-making based on these commercial tests scientifically invalid.
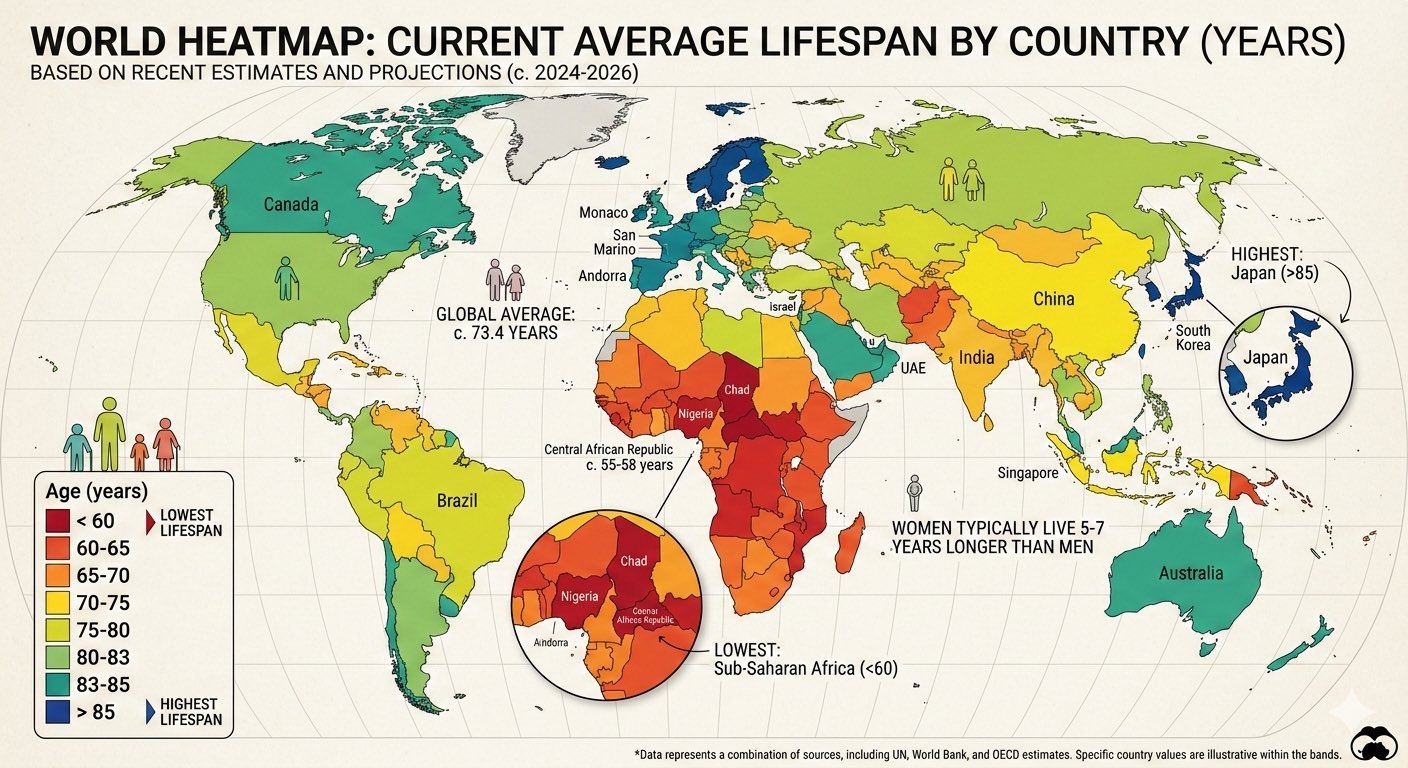
A significant portion of the analysis focuses on the macroscopic failure of the United States healthcare system to optimize healthspan. While the US excels at catastrophic care and extending the lives of older adults with established pathologies, it fails fundamentally at primary prevention. Consequently, the US has engineered an “expansion of morbidity,” where lifespan is artificially propped up by medical technology, but healthspan remains stagnant or degrades. US life expectancy peaked in 2014 and currently ranks in the 60s globally, trailing behind nations like South Korea, Singapore, and Japan. Crimmins argues that the most potent levers for biological aging are not found in isolated molecular interventions deployed in late adulthood, but rather in the mitigation of early-life social stressors, childhood infectious burdens, and persistent behavioral liabilities (obesity, physical inactivity, and substance abuse) that hardwire accelerated epigenetic aging long before clinical symptoms manifest.
II. Insight Bullets
- Unified Disease Origin: Age-related pathologies (cognitive decline, cardiovascular disease, cancer) share a common origin rooted in a specific set of molecular and cellular degradations, rather than operating as independent systemic failures.
- Epigenetic vs. Chronological Age: There is a proven, quantifiable divergence between chronological age and biological age, verifiable across large human population samples.
- Epigenetic Clock Volatility: Commercial epigenetic age testing is highly unstable for individual longitudinal tracking; daily measurements in a single subject can yield a variance of 8 to 10 years.
- Biomarker Correlation: Traditional biological age indices (composites of 12+ clinical organ-system markers) correlate strongly (r 0.65 to 0.70) with modern epigenetic clocks.
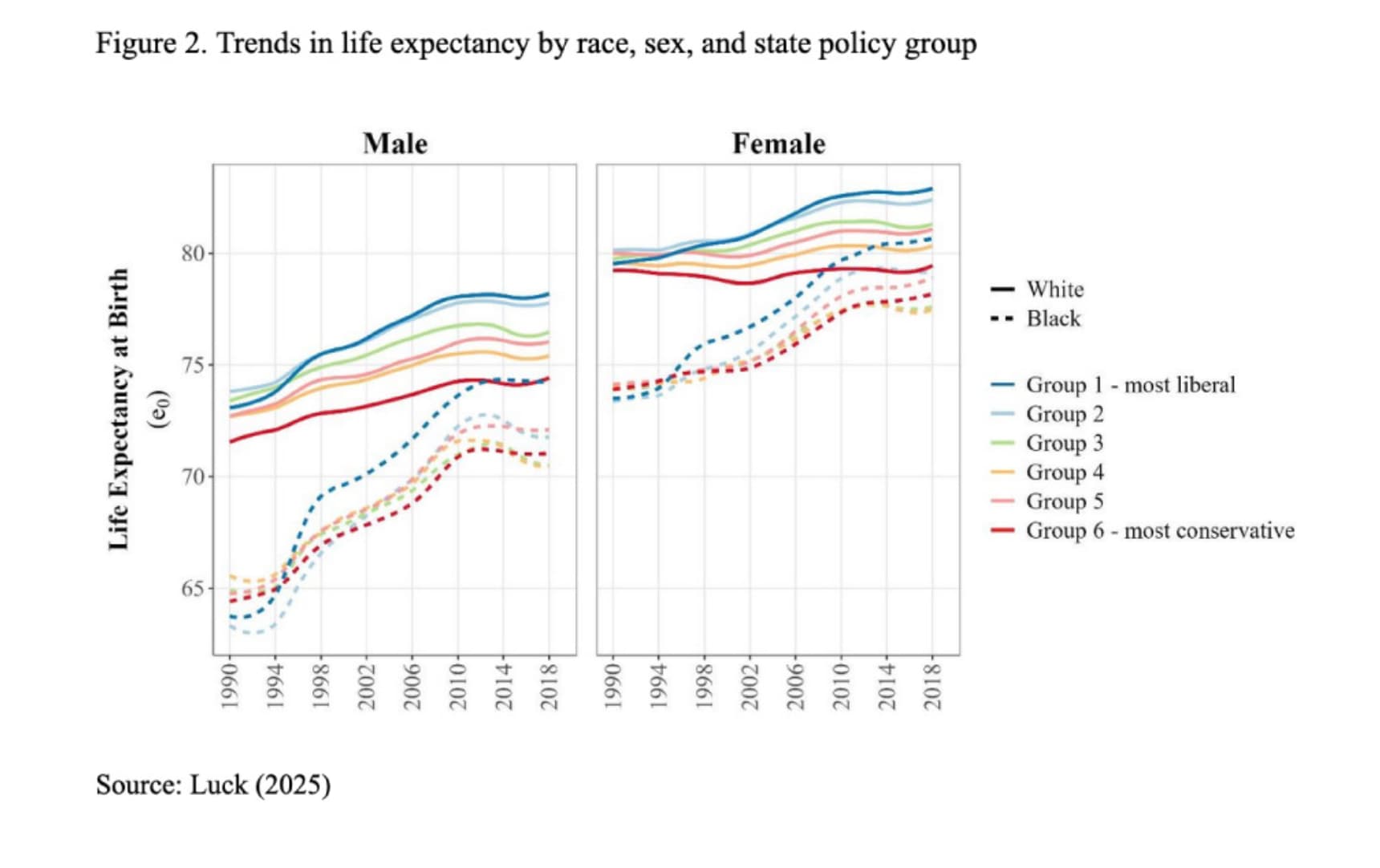
- Social Determinants as Biological Drivers: Poverty, minority status, and lack of healthcare access translate directly into accelerated molecular aging, visible in epigenetic markers by a subject’s late 20s.
- Early-Life Critical Windows: Epigenetic trajectories are heavily influenced by childhood conditions, and potentially by intergenerational stressors (e.g., maternal/grandmother environments).
- The Cohort Morbidity Phenotype: Historical increases in late-life survival were heavily driven by the reduction of childhood infectious disease exposure, yielding stronger adult cohorts.
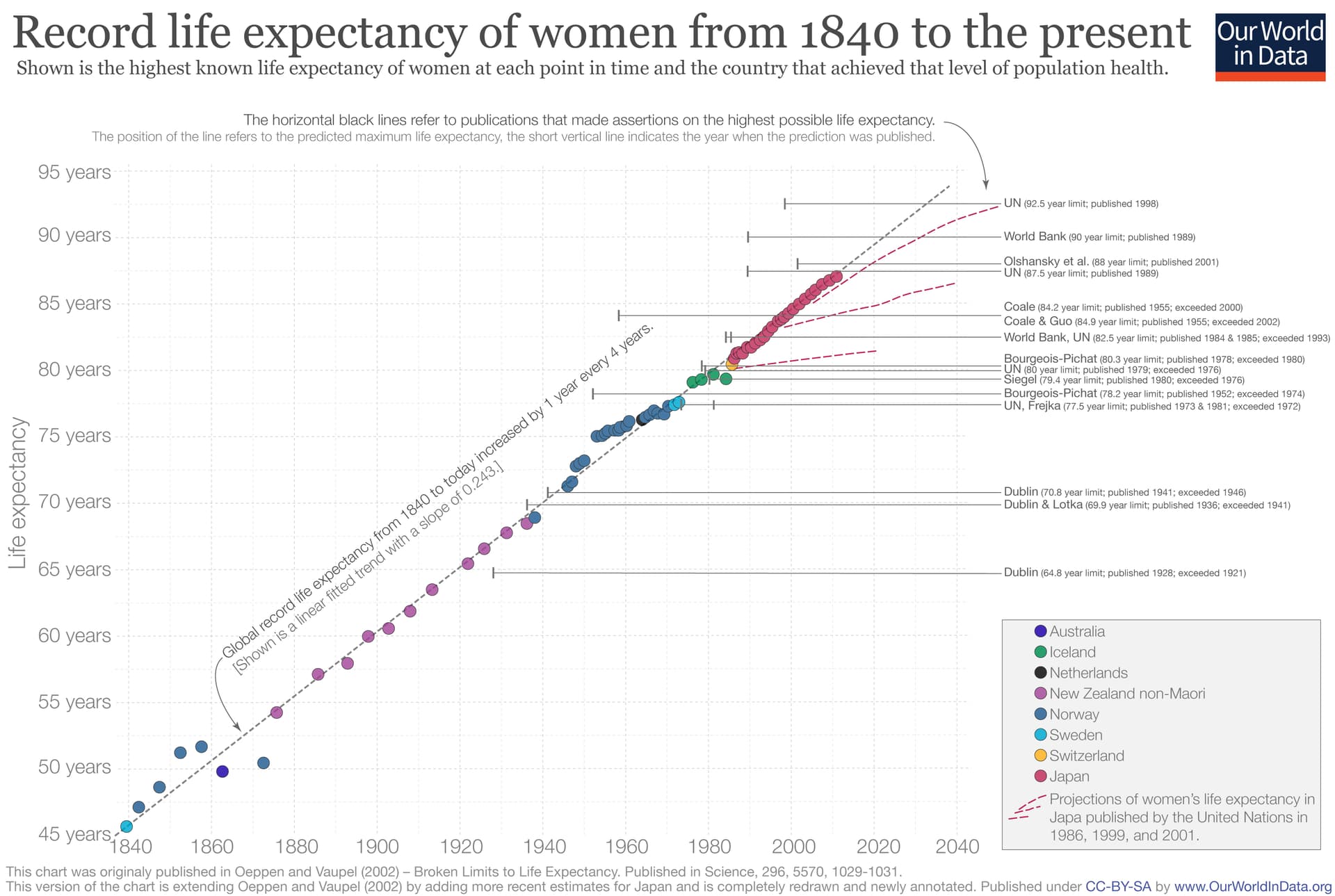
- US Life Expectancy Stagnation: US life expectancy peaked in 2014, experiencing a lost decade of progress, and now ranks in the 60s globally.
- The Lifespan/Healthspan Divergence: The US healthcare system efficiently extends the lifespan of older, diseased populations, but fails to prevent the onset of disease, leading to an expanded period of multi-morbidity.
- Demographic Reversals: South Korea, Hong Kong, and Singapore have rapidly outpaced the US in longevity, exceeding US life expectancy by approximately five years.
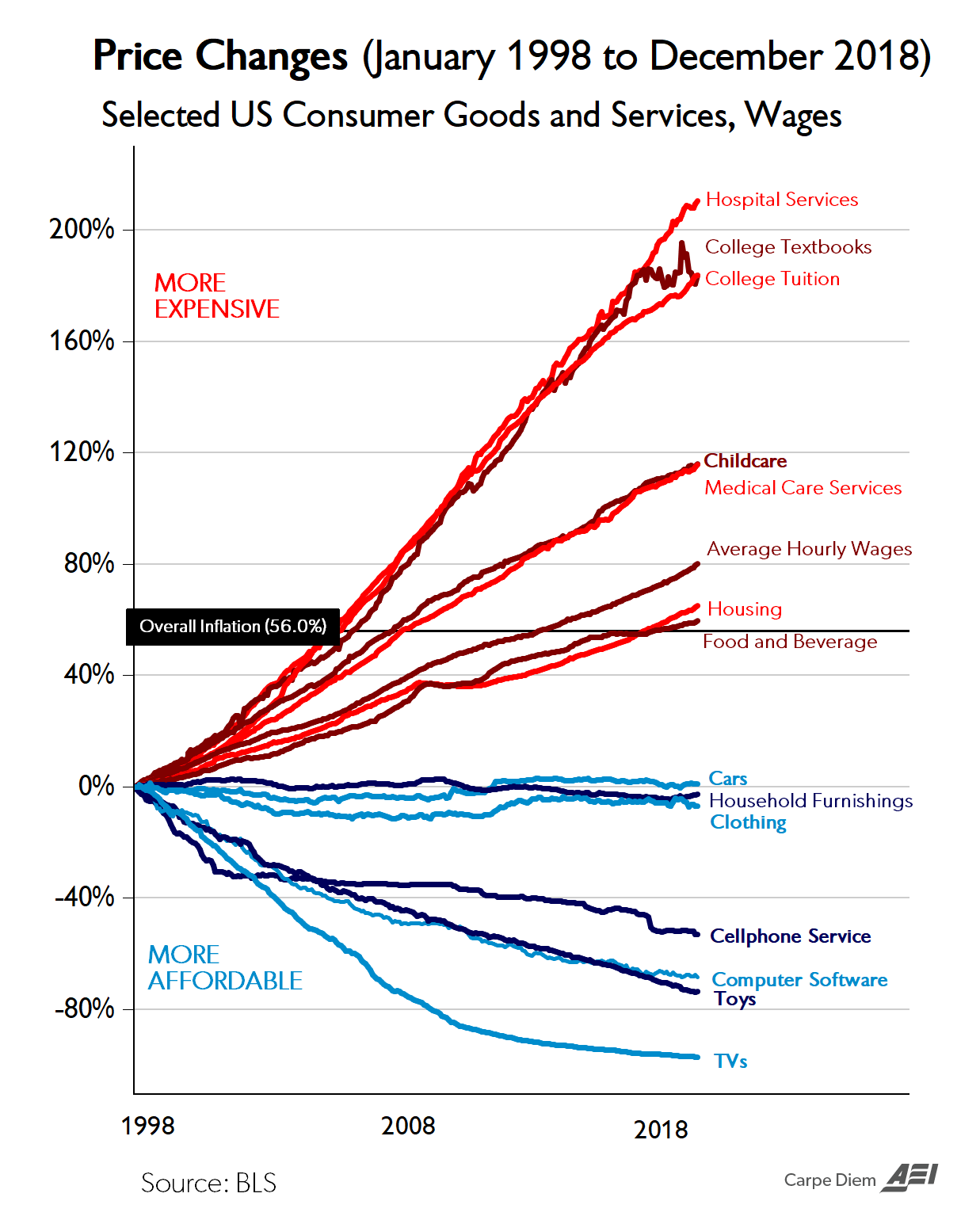
- Behavioral Liabilities: The unique US longevity deficit is heavily mediated by high caloric intake, low physical activity, and middle-age mortality shocks from the opioid/drug epidemic.
- Gender Parity in Morbidity: As global social gender equality increases, behavioral risk profiles (like smoking in European women) converge, closing the health/survival gap between sexes.
III. Adversarial Claims & Evidence Table
| Claim from Video | Speaker’s Evidence | Scientific Reality (Current Data) | Evidence Grade | Verdict |
|---|---|---|---|---|
| Epigenetic age can fluctuate 8-10 years when measured daily in one person. | Recent un-cited meeting presentation. | First-generation epigenetic clocks (e.g., Horvath 2013) suffer from significant technical noise. Next-generation principal component (PC) clocks and rate-of-aging clocks (DunedinPACE) have reduced this variance, but technical and biological noise in consumer tests remains a major barrier to individual tracking. | Level B | Plausible |
| Traditional clinical biological age correlates ~0.65-0.70 with epigenetic age. | Personal/Institutional cohort data. | Validated. Studies comparing composite clinical biomarkers (like PhenoAge) with DNA methylation clocks show moderate to strong correlations, confirming they track overlapping but distinct biological pathways. (Levine et al., 2018) | Level C | Strong Support |
| US life expectancy ranks in the 60s globally and peaked in 2014. | Demographic data. | Accurate. According to the UN Population Division and WHO, US life expectancy peaked around 2014 (78.9 years) and subsequently dropped due to cardiometabolic disease, COVID-19, and the opioid epidemic, currently ranking behind ~60 other nations. (CDC NCHS Data) | Level A | Strong Support |
| Early life infections dictate late-life mortality (Cohort Morbidity Phenotype). | Research with Caleb Finch. | Strongly supported by historical demographic data. Reduced inflammatory burden from childhood infections correlates with reduced cardiovascular and neurodegenerative disease decades later. (Finch & Crimmins, 2004) | Level C | Strong Support |
| Silicon Valley “biohacking” substances lack proof for delaying human aging. | Expert opinion/observational critique. | Accurate. While pre-clinical models show lifespan extension via rapamycin, senolytics, and NAD+ precursors, there are zero completed human RCTs proving lifespan extension or definitive epigenetic reversal in healthy populations. | Level A (Absence of Data) | Strong Support |
| Social hardships in childhood accelerate epigenetic aging by the late 20s. | Cohort studies (e.g., Health and Retirement Study). | Early-life socioeconomic disadvantage, trauma, and systemic discrimination are robustly associated with accelerated DNA methylation aging and telomere attrition in young adulthood. (Simons et al., 2021) | Level C | Strong Support |
IV. Actionable Protocol (Prioritized)
Because this transcript originates from a population health demographer rather than a clinical trialist, the “protocol” shifts from acute pharmacological interventions to systemic and environmental risk mitigation.
High Confidence Tier (Protocols backed by robust longitudinal data)
- Prioritize Standard Clinical Biomarkers over Epigenetic Clocks: Do not alter clinical protocols based on short-term changes in direct-to-consumer epigenetic age tests due to unacceptable standard deviations (8-10 year noise margins). Rely on composite, traditional physiological systems testing (e.g., advanced lipid panels, HbA1c, renal/hepatic function, VO2 max) to assess biological aging.
- Aggressive Early-Life/Adolescent Optimization: Interventions to maximize healthspan must be front-loaded. Mitigating chronic inflammation, securing metabolic health, and optimizing sleep/stress in the 20s and 30s dictates the epigenetic trajectory for the 70s.
- Behavioral Basics as Primary Biohacks: The data clearly indicates that the US longevity deficit is driven by fundamental metabolic and behavioral failures. Absolute priority must be given to matching caloric intake with daily energy expenditure (physical activity) to avoid the uniquely American obesity-driven morbidity expansion.
Experimental Tier (Mechanistically plausible but requiring long-term human validation)
- Pharmacological Biohacking: The use of targeted molecular therapies (mTOR inhibitors, senolytics) remains experimental. While they target the “hallmarks of aging” mentioned in the transcript, their deployment in healthy human populations lacks the safety and efficacy endpoints established by Level A/B evidence.
Red Flag Zone (Claims debunked or lacking safety/efficacy data)
- Short-term Epigenetic Tracking: Testing epigenetic age daily, weekly, or monthly to validate a new supplement or diet. The technical noise completely obscures the biological signal.
- Relying on Late-Life Medical Rescue: Planning to utilize the US healthcare system to preserve healthspan in late adulthood. The system is designed to extend lifespan post-disease onset, resulting in a prolonged state of morbidity rather than an extended healthspan.
V. Technical Mechanism Breakdown
- Epigenetic Age (DNA Methylation Clocks): The transcript heavily references epigenetic aging. This refers to the addition of methyl groups (CH3) to cytosine bases in the DNA sequence, specifically at CpG dinucleotides. These methylation patterns dictate gene expression (turning genes on or off) without altering the underlying genetic code. As organisms age, highly predictable shifts in methylation occur (hypermethylation of tumor suppressor genes, global hypomethylation). Clocks developed by Steve Horvath and others use machine learning to correlate these methylation patterns with chronological age and mortality risk.
- Expansion of Morbidity vs. Compression of Morbidity: The transcript outlines a failure to achieve “compression of morbidity.” In an ideal gerontological scenario, human lifespan approaches its maximum limit while the onset of chronic disease is delayed until the very end of life, compressing the period of illness into a brief window. Instead, the US is experiencing “expansion of morbidity,” where medical technology (e.g., statins, advanced oncology, stents) artificially suppresses the mortality rate of chronic diseases without addressing the root cellular senescence. The result is a prolonged, decades-long survival phase characterized by compounding polypharmacy and multi-morbidity.
- Allostatic Load and Biological Weathering: The biological translation of the “social factors” discussed by Crimmins occurs via the HPA (hypothalamic-pituitary-adrenal) axis and sympathetic nervous system. Chronic psychological or socioeconomic stress leads to persistent cortisol elevation and systemic low-grade inflammation. This sustained “allostatic load” accelerates cellular aging mechanisms, specifically driving telomere attrition and inducing premature cellular senescence, visually quantified as advanced biological age in marginalized demographics.