Wish the commercial price on Amazon was more palatable. Too much on my stack to take before it all expires. Does anyone have experience with this?
In-depth review reveals dietary lipid intervention as potential strategy to prevent brain aging
Dietary Lipid Intervention in the Prevention of Brain Aging
The review focuses on the intricate relationship between lipid homeostasis and brain aging, emphasizing the importance of maintaining synaptic plasticity to prevent cognitive impairment. By examining evidence from epidemiological and animal studies, the research team demonstrates the crucial functions of polyunsaturated fatty acids (PUFAs), specifically phospholipids such as phosphatidylcholine, phosphatidylserine, and plasmalogen, in promoting brain health.
Full (Open access) paper:
https://www.sciencedirect.com/science/article/pii/S2095809923002709?via%3Dihub
A new option in the plasmalogen supplement market:
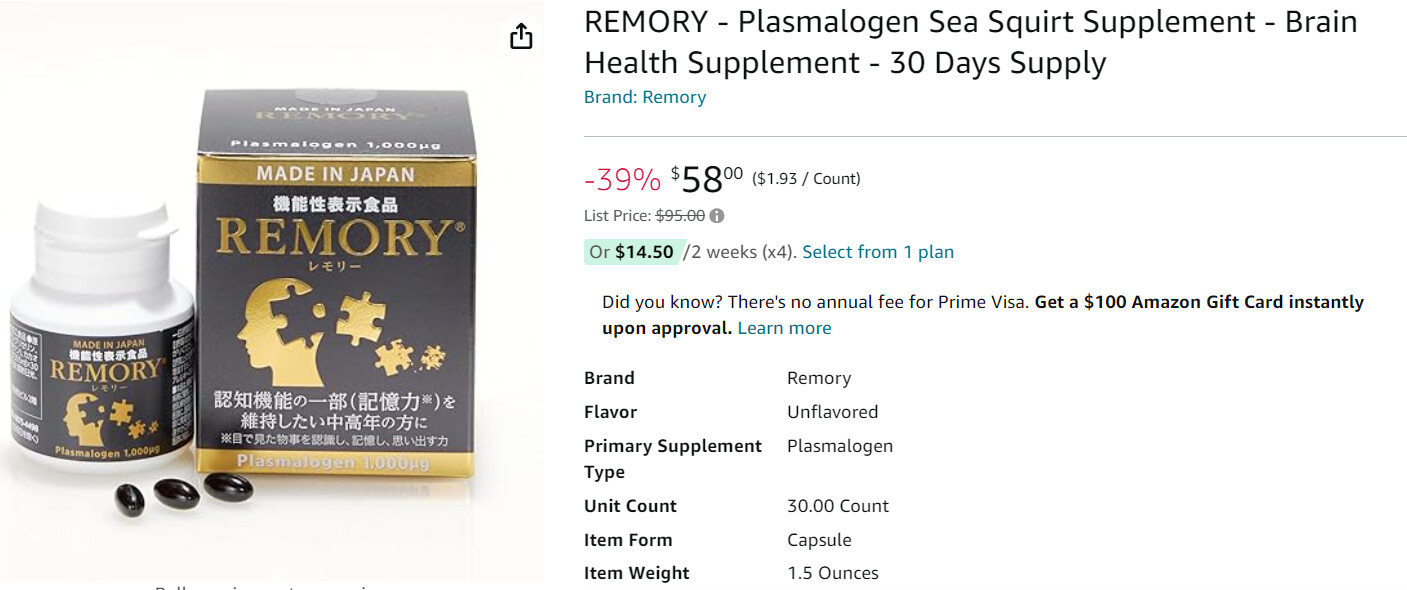
REMORY: Elevate Cognitive Health with Unique Sea Squirt-Derived Plasmalogens. Now in the U.S., this Japanese-Innovated Supplement Combines EPA, DHA, and Omega-3s
LOS ANGELES, CA / ACCESSWIRE / November 16, 2023 / REMORY®, a supplement known for containing plasmalogens, which are recognized for their role in preventing cognitive decline, has been launched in the United States. Since its introduction in October 2023, the product has received considerable attention.
Hokkori USA, a distributor known for its commitment to quality and customer satisfaction, has recently announced its role as the authorized distributor of a health product called REMORY®, a supplement known for containing plasmalogens, which are recognized for their role in preventing cognitive decline manufactured by Moolint, Inc.
REMORY® contains plasmalogens, components also found in the human brain, which play a crucial role in maintaining cognitive function. This supplement is particularly suitable for middle-aged and older adults who experience a decline in memory. Unlike other plasmalogens, REMORY® utilizes plasmalogens derived from sea squirts (ascidians), a source known for its rich content of not only plasmalogens but also EPA, DHA, and Omega-3 fatty acids. The primary ingredient in REMORY® is high-quality sea squirts sourced entirely from Hokkaido, Japan.
Furthermore, REMORY® is the only sea squirt-derived plasmalogen supplement in the U.S. market that has been granted permission by the Japanese Government to be labeled as a “Food with Functional Claims.”
Japan, facing rapid aging compared to other countries globally, has a high demand for products that help maintain memory, and many seniors have experienced the benefits of plasmalogens.
Interestingly sea squirts are one of the few animals to use vanadium-containing proteins.
Vanadium has been reported to lower blood glucose in various diabetic animal models.
2 dollars a day for this. If it works, then it’s probably worth it for me. If it doesn’t maybe I’ll forget the whole thing.
I’ve been using the product below for over a year. Same dosage. Lower cost.
https://www.amazon.com/REMORY-Plasmalogen-Squirt-Supplement-Health/dp/B0CGY6T59K
1,000 ug (1 mg) per capsule.
Pardon the error. That was in reply to RapAdmin.
What do you feel are your personal (albeit n=1) benefits from it? What is your age? Thanks!
Recommended by Dave Asprey (vanadyl sulfate).
I’m not sure the use of vanadyl sulfate is particularly new, I remember reading about it 30 years ago.
Researchers found the long form - but not the short one - in the so-called endfeet of astrocytes. Astrocytes are a kind of support cell that help maintain the barrier between the brain and the rest of the body. Their endfeet wrap around tiny blood vessels in the brain and help regulate blood flow. Astrocytic endfeet are the perfect place to be if your job is to keep the brain free of unwanted proteins by flushing waste out of the brain and into the bloodstream, where it can be carried away and disposed of.
Thinking that increasing the amount of long aquaporin 4 might increase waste clearance, researchers screened 2,560 compounds for the ability to increase readthrough of the aquaporin 4 gene. They found two: apigenin, a dietary flavone, and [sulphaquinoxaline]
(Sulfaquinoxaline | C14H12N4O2S | CID 5338 - PubChem), a veterinary antibiotic. Sulphaquinoxaline is not safe for use in people. Apigenin is available as a dietary supplement, but it’s not known how much gets into the brain. The researchers studied mice genetically engineered to have high levels of amyloid in their brains. They treated the mice with apigenin; sulphaquinoxaline; an inert liquid; or a placebo compound that has no effect on readthrough. Mice treated with either apigenin or sulphaquinoxaline cleared amyloid beta significantly faster than those treated with either of the two inactive substances.
$2/day for 1mg/day for Remory vs $3 or $6 per day for the Prodrome products, which have 900mg/day plasmologens.
RapaAdmin previously estimated that the dosage used in mice studies correspond to 1800mg/day for a 72kg human.
Prodrome recommends a loading dose of 1800-3600mg/day for 1-3 months followed by a maintenance dose of 900mg/day. They also recommend getting a $500+ blood test first to determine if you are even deficient and to determine if you need their $3/day product (omega-9 based Plasmalogens) or the $6/day product (omega-3 based Plasmalogens), or both.
Looks like the Prodrome $500+ blood test for Plasmalogen levels is availabe for $350 from Opencures ProdromeScan blood test from OpenCures. The slight catch is that this involves your blood sample first being sent to Opencures (near San Francisco) who keeps some of the blood in their freezer for research and then sends the rest to Prodrome (in San Diego). Sounds like a deal to me.
I tried to place an order but the website for opencures.org seems to be having issues with accepting payment. I have contacted support@opencures.org.
Yes - that sounds like a good testing program. Opencures is a relatively well known organization, started by Kevin Perrott who was on the SENS board of directors. He’s very into longevity, so I suspect his organization is a good one.
This seems like something that actually add credence- or do people know more about FFC?
“ Registering Foods with Function Claims in Japan
The Japanese government has established clear guidelines for FFC and a strict set of criteria that must be met before a product can be labeled as FFC. This regulation ensures the safety, quality, and efficacy of products marketed as having health benefits.
Each food or supplement product must be registered with CAA’s centralized system. The application and registration process must be done through CAA’s database. Successful FFC registration takes at least six months and involves numerous steps and documents.
As part of the application process, companies are required to clearly and accurately state their products’ claims and provide scientific evidence to back them up.
The following methods evaluate the efficacy of FFC:
- A clinical trial with a finished product and/or,
- Systematic literature review on a finished product or active ingredient”
Foods with Function Claims in Japan: What You Need to Know | RegASK.
Not sure how accurate this is… these “market research reports” have a tendency to be complete crap if they aren’t from a known market research group. But, perhaps a starting point if anyone is looking for other sources of plasmalogens:
Some of the major companies influencing this Plasmalogen Health Supplements market include:
• Daiwa Pharmaceutical
• The Fuji &
• B&S Corporation
• Prodrome Sciences
• CEREGAIN
• MARRIENE.Co.,Ltd
• Bio-Mer China
Doesn’t mention Moolint (makes Remory). I ordered some right away and still has not come. Don’t know what’s going on. It looks like good stuff and I wondered whether the vanadium might help my fasting glucose.
Did you order it from Amazon? Even Amazon has a 2-3 week delivery date for this product.
If you ordered elsewhere it probably will take even longer. A bit pricey.
Yes, too much money probably but I bought the cheap stuff above to tide me over till it gets here. Seems like a good product to me.